NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 51
The correct order of reactivity of the following benzyl halides towards reaction with KCN is:
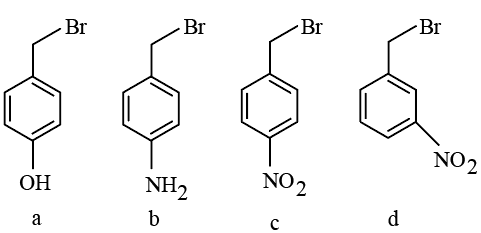
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 52
Given below are four compounds :
(a) n-propyl choride
(b) iso-propyl chloride
(c) sec-butyl chloride
(d) neo-pentyl chloride
Percentage of carbon in the one which exhibits optical isomerism is:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 53
By usual analysis, 1.00 g of compow1d (X) gave 1.79 g of magnesium pyrophosphate. The percentage of phosphorus in compound (X) is: (nearest integer)
(Given, molar mass in $$gmol^{-1}$$ : 0 = 16, Mg = 24, P = 31 )
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 54
Given below are two statements:
Statement I : Crystal Field Stabilization Energy (CFSE) of $$\left[Cr\left( H_{2}O \right)_{6} \right]^{2+}$$ is greater than that of $$\left[Mn\left( H_{2}O \right)_{6} \right]^{2+}$$.
Statement II: Potassium ferricyanide has a greater spin-only magnetic moment than sodium ferrocyanide.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 55
Aqueous HCI reacts with $$MnO_{2} \left(s\right)$$ to form $$MnCl_{2}\left(aq\right)$$, $$Cl_{2}\left(g\right)$$ and $$H_{2}O\left(l\right)$$. What is the weight (in g) of Cl_{2} liberated when 8.7 g of $$MnO_{2} \left(s\right)$$ is reacted with excess aqueous HCI solution ?
(Given Molar mass in g $$mol^{-1}$$ Mn = 55, Cl = 35.5, 0 = 16, H = l )
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 56
For a closed circuit Daniell cell, which of the following plots is the accurate one at a given temperature?
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 57
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 58
Consider the following data :
$$\Delta_f H^\ominus$$ (methane, g) = - X kJ $$mol^{-1}$$
Enthalpy of sublimation of graphite = Y kJ $$mol^{-1}$$
Dissociation enthalpy of $$H_{2}$$ = Zkj $$mol^{-1}$$
The bond enthalpy of C - H bond is given by :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 59
The correct order of the rate of the reaction for the following reaction with respect to nucleophiles is:
$$CH_{3}Br + Nu^{\ominus} \rightarrow CH_{3}Nu+Br^{\ominus}$$
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 60
Given below are two statements:
Statement I: Compound (X), shown below, dissolves in $$NaHCO_{3}$$ solution and has two chiral carbon atoms
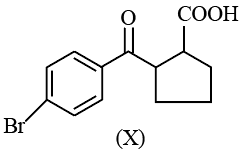
Statement II: Compound (Y), shown below, has two carbons with $$sp^{3}$$ hybridization, one carbon with $$sp^{2}$$ and one carbon with sp hybridization
%20slot%202.png)
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 61
Given below are two statements:
Statement I: The correct order in terms of bond dissociation enthalpy is $$Cl_{2} > Br_{2} > F_{2} > I_{2}$$.
Statement II : The correct trend in the covalent character of the metal halides is $$[SnCl_{4} > SnCl_{2}]$$, $$[PbCl_{4}> PbCl_{2}]$$, and $$[UF_{4} > UF_{6}]$$.
In The light oh the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 62
Consider the following spectral lines for atomic hydrogen :
A. First line of Paschen series
B. Second line of Balmer series
C. Third line of Paschen series
D. Fourth line of Bracket series
The correct arrangement of the above lines in ascending order of energy is :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 63
Match List - I with List - II.
%20slot%202.png)
Choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 64
Given below are some of the statements about Mn and $$Mn_{2}O_{7}$$. Identify the correct statements.
A. Mn forms the oxide $$Mn_{2}O_{7}$$, in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn.
C. $$Mn_{2}O_{7}$$ is an ionic oxide.
D. The structure of $$Mn_{2}O_{7}$$ consists of one bridged oxygen.
Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 65
Decompasition of A is a first order reaction at T(K) and is given by $$A(g) \rightarrow B(g)+C(g)$$.
In a closed 1 L vessel, 1 bar A(g) is allowed to decompose at T(K). After 100 minutes, the total pressure was 1.5bar. What is the rate constant $$(in min^{-1})$$ of the reaction ? (log 2 = 0.3)
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 66
The correct increasing order of C - H(A), C - 0 (B), C = O(C) and C = N (D) bonds in terms of covalent bond length is :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 67
On heating a mixture of common salt and $$K_{2}Cr_{2}O_{7}$$ in equal amount along with concentrated $$H_{2}SO_{4}$$ in a test tube, a gas is evolved. Formula of the gas evolved and oxidation State of the central metal atom in the gas respectively are:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 68
Given below are two statements:
Statement I: The correct order in terms of atomic/ionic radii is $$Al >Mg > Mg^{2+} >Al^{3+}$$
Statement II: The correct order in terms of the magnitude of electron gain enthalpy is Cl > Br >S >O.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 69
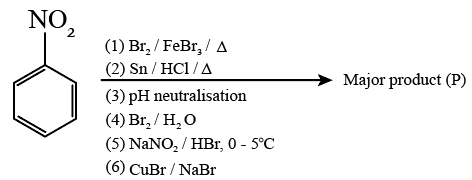
Consider the above sequence of reactions. The number of bromine atom(s) in the final product (P) will be:
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 70
The correct statements are :
A. Activation energy for enzyme catalysed hydrolysis of sucrose is lower than that of acid catalysed hydrolysis.
B. During denaturation, secondary and tertiary structures of a protein are destroyed but primary structure remains intact.
C. Nucleotides are joined together by glycosidic linkage between $$C_{1}$$ and $$C_{4}$$ carbons of the pentose sugar.
D. Quaternary structure of proteins represents overall folding of the polypeptide chain.
Choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 71
The first and second ionization constants of $$H_{2}X$$ are $$2.5 \times 10^{-8}$$ and $$1.0 \times 10^{-13}$$ respectively. The concentration of $$X^{2-}$$ in $$0.1 MH_{2} X$$ solution is _________ $$\times 10^{-15}M$$. (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 72
A substance 'X' (1.5 g) dissolved in 150 g of a solvent 'Y'(molar mass=300 g $$mol^{-1}$$ ) led to an elevation of the boiling point by 0.5 K. The relative lowering in the vapour pressure of the solvent 'Y' is __________ $$\times 10^{-2}$$. (nearest integer)
[Given : $$K_{b}$$ of the solvent =5.0 K kg $$mol^{-1}$$]
Assume the solution to be dilute and no association or dissociation of X takes place in solution.
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 73
The osmotic pressure of a living cell is 12 atm at 300 K. The strength of sodium chloride solution that is isotonic with the living cell at tltis temperature is __________ $$g L^{-1}$$.
(Nearest integer)
Given: R = 0.08 L atm $$K^{-1} mol^{-1}$$
Assume complete dissociation of NaCl
(Given : Molar mass of Na and Cl are 23 and 35.5 g $$mol^{-1}$$ respectively.)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 74
MX is a sparingly soluble salt that follows the given solubility equilibrium at 298 K.
$$MX(s) \rightleftharpoons M^{+} (aq)+X^{-}(aq); K_{sp}=10^{-10}$$
If the standard reduction potential for $$M^{+}(aq)\xrightarrow {+e^{-}}M(s)$$ and $$\left(E_{M^{+}/M}^\ominus\right)=0.79V$$, then the value of the standard reduction potential for the metal/metal insoluble salt electrode $$E^{\ominus}_{X^{-}/MX (s)/M }$$ is __________mV.(nearest integer)
[Given : $$\frac{2.303RT}{F} = 0.059V$$]
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 2 - Chemistry - Question 75
Identify the metal ions among $$Co^{2+}, Ni^{2+}, Fe^{2+}, V^{3+} \text{ and } Ti^{2+}$$ having a spin-only magnetic moment value more than 3.0 BM. The sum of unpaired electrons present in the high spin octahedral complexes formed by those metal ions is __________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




