Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The correct order of reactivity of the following benzyl halides towards reaction with KCN is:
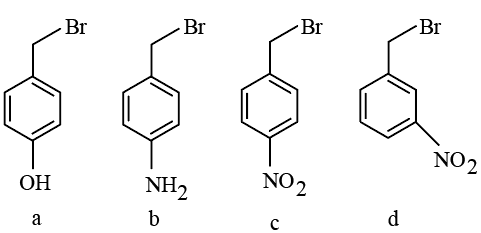
This reaction proceeds via the Sₙ1 mechanism.
The rate depends on the stability of the benzyl carbocation formed after departure of Br.
Key Idea:
Electron-donating groups increase carbocation stability and hence increase rate.
Electron-withdrawing groups decrease carbocation stability and hence decrease rate.
Resonance effects (para position) are stronger than inductive effects (meta position).
Analysis:
(b) p-NH₂
Strong +M effect, donates electron density by resonance, gives maximum stabilization → highest reactivity
(a) p-OH
Also shows +M effect but weaker than NH₂ → slightly lower reactivity
(d) m-NO₂
Shows -I effect only (no resonance at meta position) → moderate destabilization
(c) p-NO₂
Strong -M and -I effect, withdraws electron density strongly → least stable carbocation
Final Order:
b > a > d > c
Answer: p-NH₂ > p-OH > m-NO₂ > p-NO₂
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
