Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
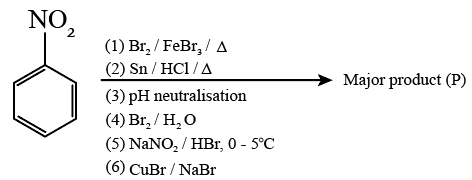
Consider the above sequence of reactions. The number of bromine atom(s) in the final product (P) will be:
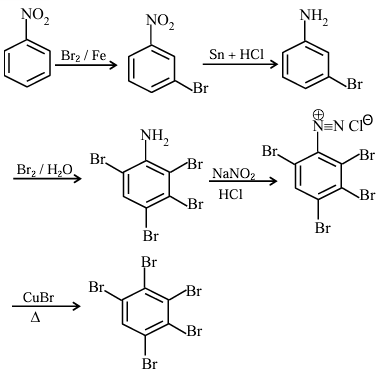
Reagent: Br₂ / FeBr₃
Reaction name: Halogenation
Product: m-bromonitrobenzene
Reagent: Sn / HCl
Reaction name: Reduction of nitro group
Product: m-bromoaniline
Reagent: Br₂ / H₂O
Reaction name: Bromination of aniline
Product: tribromoaniline derivative
Reagent: NaNO₂ / HBr (0-5°C)
Reaction name: Diazotization
Reagent: CuBr / NaBr
Reaction name: Sandmeyer reaction
Total bromine atoms = 5
Number of bromine atoms = 5
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
