NTA JEE Main 7th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 1 - Question 31
Oxidation number of potassium in K$$_2$$O, K$$_2$$O$$_2$$ and KO$$_2$$, respectively, is:
NTA JEE Main 7th January 2020 Shift 1 - Question 32
In comparison to the zeolite process for the removal of permanent hardness, the synthetic resin method is
NTA JEE Main 7th January 2020 Shift 1 - Question 33
A solution of m-chloroaniline, m-chlorophenol and m-chlorobenzoic acid in ethyl acetate was extracted initially with a saturated solution of NaHCO$$_3$$ to give fraction A. The left over organic phase was extracted with dilute NaOH solution to give fraction B. The final organic layer was labelled as fraction C. Fractions A, B and C, contain respectively:
NTA JEE Main 7th January 2020 Shift 1 - Question 34
The increasing order of pK$$_b$$ for the following compounds will be:
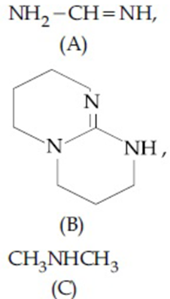
NTA JEE Main 7th January 2020 Shift 1 - Question 35
Consider the following reactions:
(a) $$(CH_3)_3CCH(OH)CH_3 \xrightarrow{\text{conc.} H_2SO_4}$$
(b) $$(CH_3)_2CHCH(Br)CH_3 \xrightarrow{\text{alc. KOH}}$$
(c) $$(CH_3)_2CHCH(Br)CH_3 \xrightarrow{(CH_3)_3O^\ominus K^\oplus}$$
(d)
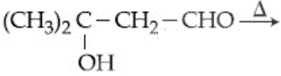
Which of the reaction(s) will not produce Saytzeff product?
NTA JEE Main 7th January 2020 Shift 1 - Question 36
At 35 $$^\circ$$C, the vapour pressure of CS$$_2$$ is 512 mmHg and that of acetone is 144 mmHg. A solution of CS$$_2$$ in acetone has a total vapour pressure of 600 mmHg. The false statement amongst the following is:
NTA JEE Main 7th January 2020 Shift 1 - Question 37
Given that the standard potentials (E$$^\circ$$) of Cu$$^{2+}$$/Cu and Cu$$^+$$/Cu are 0.34V and 0.522V respectively, the E$$^\circ$$ of Cu$$^{2+}$$/Cu$$^+$$ is:
NTA JEE Main 7th January 2020 Shift 1 - Question 38
The purest form of commercial iron is:
NTA JEE Main 7th January 2020 Shift 1 - Question 39
The atomic radius of Ag is closest to
NTA JEE Main 7th January 2020 Shift 1 - Question 40
The IUPAC name of the complex [Pt(NH$$_3$$)$$_2$$Cl(NH$$_2$$CH$$_3$$)]Cl is


