NTA JEE Main 7th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 1 - Question 41
The theory that can completely/properly explain the nature of bonding in [Ni(CO)$$_4$$] is:
NTA JEE Main 7th January 2020 Shift 1 - Question 42
1-methyl ethylene oxide when treated with an excess of HBr produces
NTA JEE Main 7th January 2020 Shift 1 - Question 43
What is the product of the following reaction?
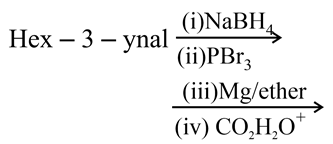
NTA JEE Main 7th January 2020 Shift 1 - Question 44
Consider the following reaction: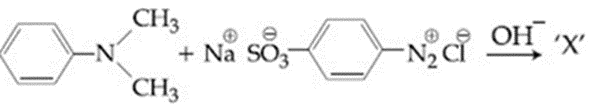
The product 'X' is used:
NTA JEE Main 7th January 2020 Shift 1 - Question 45
Match the following:
(i) Riboflavin (a) Beriberi
(ii) Thiamine (b) Scurvy
(iii) Pyridoxine (c) Cheilosis
(iv) Ascorbic acid (d) Convulsions
NTA JEE Main 7th January 2020 Shift 1 - Question 46
For the reaction:
A(l) $$\rightarrow$$ 2B(g)
$$\Delta U = 2.1$$ kcal, $$\Delta S = 20$$ cal K$$^{-1}$$ at 300K.
Hence $$\Delta G$$ in kcal is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Question 47
Two solutions, A and B, each of 100L was made by dissolving 4g of NaOH and 9.8g of H$$_2$$SO$$_4$$ in water, respectively. The pH of the resultant solution obtained from mixing 40L of solution A and 10L of solution B is (log 2 = 0.3)
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Question 48
During the nuclear explosion, one of the products is $$^{90}$$Sr with half life of 6.93 years. If 1$$\mu$$g of $$^{90}$$Sr was absorbed in the bones of a newly born baby in place of Ca, how much time, in years, is required to reduce it by 90% if it is not lost metabolically
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Question 49
Chlorine reacts with hot and concentrated NaOH and produces compounds (X) and (Y). Compound (X) gives white precipitate with silver nitrate solution. The average bond order between Cl and O atoms in (Y) is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Question 50
The number of chiral carbons in chloramphenicol is
789
456
123
0.-
Clear All


