NTA JEE Main 29th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 2 - Question 41
Given below are the critical temperatures of some of the gases:
| Gas | Critical temperature (K) |
|---|---|
| He | 5.2 |
| $$CH_4$$ | 190 |
| $$CO_2$$ | 304.2 |
| $$NH_3$$ | 405.5 |
The gas showing least adsorption on a definite amount of charcoal is
NTA JEE Main 29th July 2022 Shift 2 - Question 42
In liquation process used for tin (Sn), the metal
NTA JEE Main 29th July 2022 Shift 2 - Question 43
Dinitrogen is a robust compound, but reacts at high altitude to form oxides. The oxide of nitrogen that can damage plant leaves and retard photosynthesis is
NTA JEE Main 29th July 2022 Shift 2 - Question 44
Which of the following 3d-metal ion will give the lowest enthalpy of hydration $$\Delta_{hyd}H$$ when dissolved in water?
NTA JEE Main 29th July 2022 Shift 2 - Question 45
Octahedral complexes of copper(II) undergo structural distortion (Jahn-Teller). Which one of the given copper(II) complexes will show the maximum structural distortion? (en = ethylenediamine; $$H_2N-CH_2-CH_2-NH_2$$)
NTA JEE Main 29th July 2022 Shift 2 - Question 46
Compound 'A' undergoes following sequence of reactions to give compound 'B'. The correct structure and chirality of compound 'B' is [where Et is $$-C_2H_5$$]
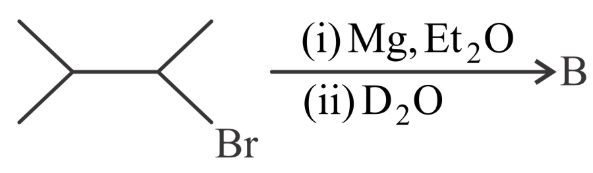
NTA JEE Main 29th July 2022 Shift 2 - Question 47
When ethanol is heated with conc. $$H_2SO_4$$, a gas is produced. The compound formed, when this gas is treated with cold dilute aqueous solution of Baeyer's reagent, is
NTA JEE Main 29th July 2022 Shift 2 - Question 48
The Hinsberg reagent is
NTA JEE Main 29th July 2022 Shift 2 - Question 49
Which of the following is NOT a natural polymer?
NTA JEE Main 29th July 2022 Shift 2 - Question 50
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Amylose is insoluble in water.
Reason R: Amylose is a long linear molecule with more than 200 glucose units.
In the light of the above statements, choose the correct answer from the options given below.


