NTA JEE Main 29th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 2 - Question 51
A 1.84 mg sample of polyhydric alcoholic compound 'X' of molar mass 92.0 g/mol gave 1.344 mL of $$H_2$$ gas at STP. The number of alcoholic hydrogen present in compound 'X' is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 52
Consider, $$PF_5, BrF_5, PCl_3, SF_6, ICl_4^-, ClF_3$$ and $$IF_5$$. Amongst the above molecule(s)/ion(s), the number of molecule(s)/ion(s) having $$sp^3d^2$$ hybridisation is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 53
'x' g of molecular oxygen $$(O_2)$$ is mixed with 200 g of neon (Ne). The total pressure of the non-reactive mixture of $$O_2$$ and Ne in the cylinder is 25 bar. The partial pressure of Ne is 20 bar at the same temperature and volume. The value of 'x' is [Given: Molar mass of $$O_2$$ = 32 g mol$$^{-1}$$. Molar mass of Ne = 20 g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 54
1.80 g of solute A was dissolved in 62.5 cm$$^3$$ of ethanol and freezing point of the solution was found to be 155.1 K. The molar mass of solute A is _____ g mol$$^{-1}$$. [Given: Freezing point of ethanol is 156.0 K. Density of ethanol is 0.80 g cm$$^{-3}$$. Freezing point depression constant of ethanol is 2.00 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 55
For a cell, $$Cu(s)|Cu^{2+}(0.001 M)||Ag^+(0.01 M)|Ag(s)$$, the cell potential is found to be 0.43 V at 298 K. The magnitude of standard electrode potential for $$Cu^{2+}|Cu$$ is _____ $$\times 10^{-2}$$ V. Given: $$E^\theta_{Ag^+/Ag} = 0.80$$ V and $$\frac{2.303RT}{F} = 0.06$$ V
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 56
Assuming 1 $$\mu$$g of trace radioactive element X with a half life of 30 years is absorbed by a growing tree. The amount of X remaining in the tree after 100 years is _____ $$\times 10^{-1}$$ $$\mu$$g. [Given: $$\ln 10 = 2.303$$; $$\log 2 = 0.30$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 57
Consider the following sulphur based oxoacids. $$H_2SO_3, H_2SO_4, H_2S_2O_8$$ and $$H_2S_2O_7$$. Amongst these oxoacids, the number of those with peroxo (O-O) bond is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 58
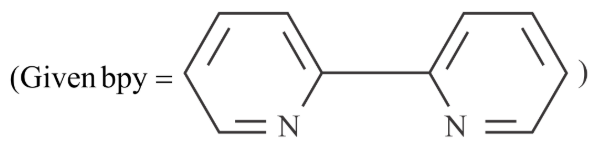
Sum of oxidation state (magnitude) and coordination number of cobalt in $$Na[Co(bpy)Cl_4]$$ is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 59
The number of stereoisomers formed in a reaction of $$[\pm] PhC(=O)C(OH)(CN)Ph$$ with HCN is
789
456
123
0.-
Clear All
NTA JEE Main 29th July 2022 Shift 2 - Question 60
The number of chlorine atoms in bithionol is
789
456
123
0.-
Clear All


