NTA JEE Main 29th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 2 - Question 31
Consider the reaction
$$4HNO_3(l) + 3KCl(s) \to Cl_2(g) + NOCl(g) + 2H_2O(g) + 3KNO_3(s)$$
The amount of $$HNO_3$$ required to produce 110.0 g of $$KNO_3$$ is (Given: Atomic masses of H, O, N and K are 1, 16, 14 and 39, respectively.)
NTA JEE Main 29th July 2022 Shift 2 - Question 32
$$C(s) + O_2(g) \to CO_2(g) + 400$$ kJ
$$C(s) + \frac{1}{2}O_2(g) \to CO(g) + 100$$ kJ
When coal of purity 60% is allowed to burn in presence of insufficient oxygen, 60% of carbon is converted into 'CO' and the remaining is converted into '$$CO_2$$'. The heat generated when 0.6 kg of coal is burnt is
NTA JEE Main 29th July 2022 Shift 2 - Question 33
Given below are the quantum numbers for 4 electrons.
A. $$n=3, l=2, m_l=1, m_s=+\frac{1}{2}$$
B. $$n=4, l=1, m_l=0, m_s=+\frac{1}{2}$$
C. $$n=4, l=2, m_l=-2, m_s=-\frac{1}{2}$$
D. $$n=3, l=1, m_l=-1, m_s=+\frac{1}{2}$$
The correct order of increasing energy is
NTA JEE Main 29th July 2022 Shift 2 - Question 34
200 mL of 0.01 M HCl is mixed with 400 mL of 0.01 M $$H_2SO_4$$. The pH of the mixture is
NTA JEE Main 29th July 2022 Shift 2 - Question 35
A compound 'X' is a weak acid and it exhibits colour change at pH close to the equivalence point during neutralization of NaOH with $$CH_3COOH$$. Compound 'X' exists in ionized form in basic medium. The compound 'X' is
NTA JEE Main 29th July 2022 Shift 2 - Question 36
Given below are two statements.
Statement I: Stannane is an example of a molecular hydride.
Statement II: Stannane is a planar molecule.
In the light of the above statement, choose the most appropriate answer from the options given below
NTA JEE Main 29th July 2022 Shift 2 - Question 37
Portland cement contains 'X' to enhance the setting time. What is 'X'?
NTA JEE Main 29th July 2022 Shift 2 - Question 38
When borax is heated with CoO on a platinum loop, blue coloured bead formed is largely due to
NTA JEE Main 29th July 2022 Shift 2 - Question 39
Correct structure of $$\gamma$$-methylcyclohexane carbaldehyde is
NTA JEE Main 29th July 2022 Shift 2 - Question 40
Given below are two statements.
Statement I: The compound is optically active.
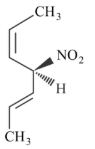
Statement II: The second compound is mirror image of the above compound A.
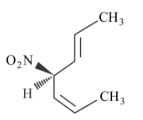
In the light of the above statement, choose the most appropriate answer from the options given below.


