NTA JEE Main 25th July 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 31
The spin only magnetic moments (in BM) for free Ti$$^{3+}$$, V$$^{2+}$$ and Sc$$^{3+}$$ ions respectively are (At.No. Sc : 21, Ti : 22, V : 23)
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 32
The ionic radii of F$$^-$$ and O$$^{2-}$$ respectively are 1.33 A and 1.4A, while the covalent radius of N is 0.74A. The correct statement for the ionic radius of N$$^{3-}$$ from the following is:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 33
In the following the correct bond order sequence is:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 34
Identify the species having one $$\pi$$-bond and maximum number of canonical forms from the following:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 35
Identify the process in which change in the oxidation state is five:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 36
Which one of the following metals forms interstitial hydride easily?
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 37
Match List I with List II:
List-I (Elements) List-II (Properties)
(a) Li (i) Poor water solubility of I$$^-$$ salt
(b) Na (ii) Most abundant element in cell fluid
(c) K (iii) Bicarbonate salt used in fire extinguisher
(d) Cs (iv) Carbonate salt decomposes easily on heating
Choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 38
The correct decreasing order of densities of the following compounds is:
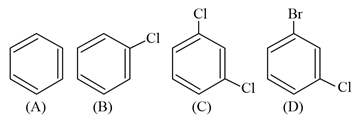
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 39
Which among the following is the strongest acid?
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 40
Given below are two statements:
Statement I : Chlorofluoro carbons breakdown by radiation in the visible energy region and release chlorine gas in the atmosphere which then reacts with stratospheric ozone.
Statement II : Atmospheric ozone reacts with nitric oxide to give nitrogen and oxygen gases, which add to the atmosphere.
For the above statements choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 41
Match List I with List II:
List-I (Example of colloids) List-II (Classification)
(a) Cheese (i) dispersion of liquid in liquid
(b) Pumice stone (ii) dispersion of liquid in gas
(c) Hair cream (iii) dispersion of gas in solid
(d) Cloud (iv) dispersion of liquid in solid
Choose the most appropriate answer from the options given below:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 42
Match List I with List II: (Both having metallurgical terms)
List-I List-II
(a) Concentration of Ag ore (i) Reverberatory furnace
(b) Blast furnace (ii) Pig iron
(c) Blister copper (iii) Leaching with dilute NaCN solution
(d) Froth floatation method (iv) Sulfide ores
Choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 43
Which one of the following metal complexes is most stable?
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 44
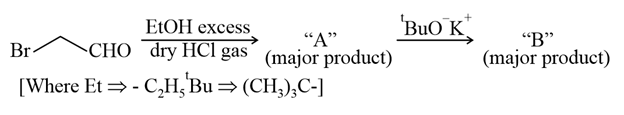
Consider the above reaction sequence, Product "A" and Product "B" formed respectively are:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 45
What is the major product "P" of the following reaction?
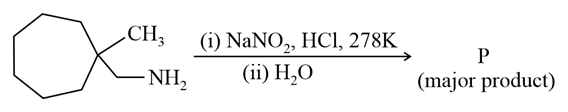
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 46
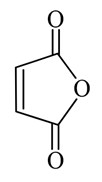
Maleic anhydride
Maleic anhydride can be prepared by:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 47
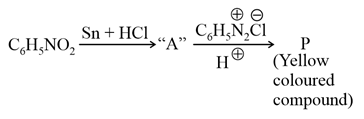
Consider the above reaction, the Product "P" is:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 48
A reaction of benzonitrile with one equivalent CH$$_3$$MgBr followed by hydrolysis produces a yellow liquid "P". The compound "P" will give positive _________ test.
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 49
A biodegradable polyamide can be made from:
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 50
Which one of the following is correct structure for cytosine?
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 51
The number of significant figures in 0.00340 is _________
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 52
An accelerated electron has a speed of $$5 \times 10^6$$ ms$$^{-1}$$ with an uncertainty of 0.02%. The uncertainty in finding its location while in motion is $$x \times 10^{-9}$$ m. The value of $$x$$ is _________. (Nearest integer)
[Use mass of electron = $$9.1 \times 10^{-31}$$ kg, h = $$6.63 \times 10^{-34}$$ Js, $$\pi$$ = 3.14]
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 53
An LPG cylinder contains gas at a pressure of 300 kPa at 27°C. The cylinder can withstand the pressure of $$1.2 \times 10^6$$ Pa. The room in which the cylinder is kept catches fire. The minimum temperature at which the bursting of cylinder will take place is _________ °C. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 54
A system does 200 J of work and at the same time absorbs 150 J of heat. The magnitude of the change in internal energy is _________J. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 55
Assuming that Ba(OH)$$_2$$ is completely ionised in aqueous solution under the given conditions the concentration of H$$_3$$O$$^+$$ ions in 0.005M aqueous solution of Ba(OH)$$_2$$ at 298 K is _________ $$\times 10^{-12}$$ mol L$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 56
0.8 g of an organic compound was analysed by Kjeldahl's method for the estimation of nitrogen. If the percentage of nitrogen in the compound was found to be 42%, then _________ mL of 1M H$$_2$$SO$$_4$$ would have been neutralized by the ammonia evolved during the analysis.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 57
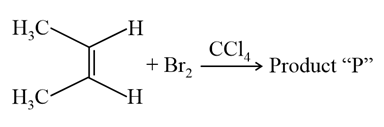
Consider the above chemical reaction. The total number of stereoisomers possible for Product "P" is _________
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 58
When 3.00 g of a substance X' is dissolved in 100 g of CCl$$_4$$, it raises the boiling point by 0.60 K. The molar mass of the substance 'X' is _________ g mol$$^{-1}$$. (Nearest integer).
[Given K$$_b$$ for CCl$$_4$$ is 5.0 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 59
For a chemical reaction A $$\rightarrow$$ B, it was found that concentration of B is increased by 0.2 mol L$$^{-1}$$ in 30 min. The average rate of the reaction is _________ $$\times 10^{-1}$$ mol L$$^{-1}$$ h$$^{-1}$$. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Chemistry - Question 60
Number of electrons present in 4f orbital of Ho$$^{3+}$$ ion is _________ (Given Atomic No. of Ho = 67)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




