NTA JEE Main 25th February 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 31
Complete combustion of 1.80 g of an oxygen containing compound ($$C_xH_yO_z$$) gave 2.64 g of $$CO_2$$ and 1.08 g of $$H_2O$$. The percentage of oxygen in the organic compound is:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 32
The plots of radial distribution functions for various orbitals of hydrogen atom against 'r' are given below. The correct plot for 3s orbital is:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 33
According to molecular orbital theory, the species among the following that does not exist is:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 34
The solubility of AgCN in a buffer solution of pH = 3 is x. The value of x is: [Assume: No cyano complex is formed; $$K_{sp}(AgCN) = 2.2 \times 10^{-16}$$ and $$K_a(HCN) = 6.2 \times 10^{-10}$$]
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 35
Which of the following equation depicts the oxidizing nature of $$H_2O_2$$?
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 36
The incorrect statement about $$B_2H_6$$ is:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 37
Compound(s) which will liberate carbon dioxide with sodium bicarbonate solution is/are:
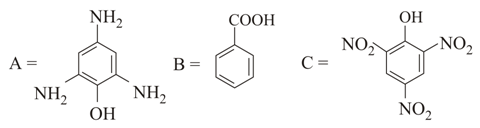
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 38
Identify A in the given chemical reaction.
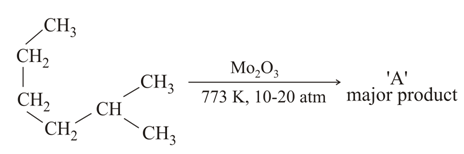
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 39
Which of the following reaction/s will not give $$p$$-aminoazobenzene?
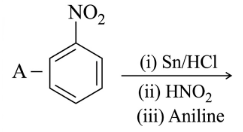
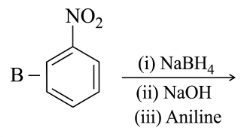
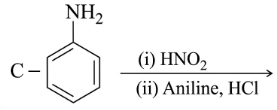
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 40
Identify A and B in the chemical reaction.
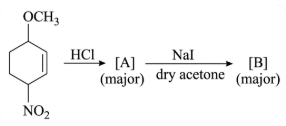
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 41
Given below are two statements:
Statement I: An allotrope of oxygen is an important intermediate in the formation of reducing smog.
Statement II: Gases such as oxides of nitrogen and sulphur present in troposphere contribute to the formation of photochemical smog.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 42
In Freundlich adsorption isotherm at moderate pressure, the extent of adsorption $$\left(\frac{x}{m}\right)$$ is directly proportional to $$P^x$$. The value of $$x$$ is:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 43
Ellingham diagram is a graphical representation of:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 44
Given below are two statements:
Statement I: $$CeO_2$$ can be used for oxidation of aldehydes and ketones.
Statement II: Aqueous solution of $$EuSO_4$$ is a strong reducing agent.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 45
In which of the following pairs, the outermost electronic configuration will be the same?
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 46
The hybridization and magnetic nature of $$[Mn(CN)_6]^{4-}$$ and $$[Fe(CN)_6]^{3-}$$, respectively are:
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 47
Which one of the following reactions will not form acetaldehyde?
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 48
The major product of the following chemical reaction is:
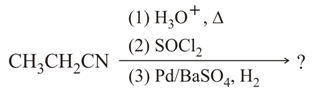
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 49
Which statement is correct?
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 50
Which of the glycosidic linkage between galactose and glucose is present in lactose?
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 51
A car tyre is filled with nitrogen gas at 35 psi at 27°C. It will burst if pressure exceeds 40 psi. The temperature in °C at which the car tyre will burst is ______ (Rounded-off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 52
The reaction of cyanamide, $$NH_2CN_{(s)}$$ with oxygen was run in a bomb calorimeter and $$\Delta U$$ was found to be $$-742.24$$ kJ mol$$^{-1}$$. The magnitude of $$\Delta H_{298}$$ for the reaction
$$NH_2CN_{(s)} + \frac{3}{2}O_{2(g)} \to N_{2(g)} + O_{2(g)} + H_2O_{(l)}$$ is ______ kJ. (Rounded off to the nearest integer) [Assume ideal gases and R = 8.314 J mol$$^{-1}$$ K$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 53
The ionization enthalpy of Na$$^+$$ formation from $$Na_{(g)}$$ is 495.8 kJ mol$$^{-1}$$, while the electron gain enthalpy of Br is $$-325.0$$ kJ mol$$^{-1}$$. Given the lattice enthalpy of NaBr is $$-728.4$$ kJ mol$$^{-1}$$. The energy for the formation of NaBr ionic solid is $$(-) \underline{\hspace{1cm}} \times 10^{-1}$$ kJ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 54
0.4 g mixture of NaOH, $$Na_2CO_3$$ and some inert impurities was first titrated with $$\frac{N}{10}$$ HCl using phenolphthalein as an indicator, 17.5 mL of HCl was required at the end point. After this methyl orange was added and titrated. 1.5 mL of same HCl was required for the next end point. The weight percentage of $$Na_2CO_3$$ in the mixture is ______ (Rounded-off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 55
In basic medium $$CrO_4^{2-}$$ oxidises $$S_2O_3^{2-}$$ to form $$SO_4^{2-}$$ and itself changes into $$Cr(OH)_4^-$$. The volume of 0.154 M $$CrO_4^{2-}$$ required to react with 40 mL of 0.25 M $$S_2O_3^{2-}$$ is ______ mL. (Rounded-off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 56
Using the provided information in the following paper chromatogram:
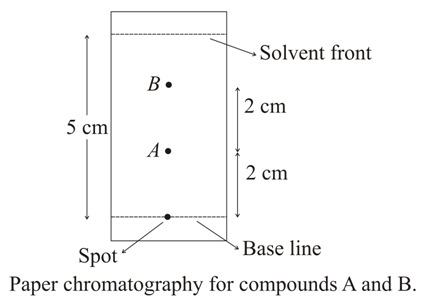
The calculated $$R_f$$ value of A is ______ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 57
Consider the following chemical reaction.
$$CH \equiv CH \xrightarrow[2) CO, HCl, AlCl_3]{1) \text{Red hot Fe tube, 873 K}}$$ Product
The number of $$sp^2$$ hybridized carbon atom(s) present in the product is ______
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 58
1 molal aqueous solution of an electrolyte $$A_2B_3$$ is 60% ionised. The boiling point of the solution at 1 atm is ______ K. (Rounded-off to the nearest integer) [Given $$K_b$$ for $$H_2O = 0.52$$ K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 59
For the reaction, $$aA + bB \to cC + dD$$, the plot of log k v/s $$\frac{1}{T}$$ is given below:
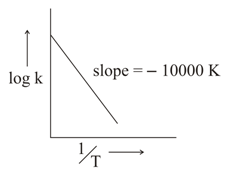
The temperature at which the rate constant of the reaction is $$10^{-4}$$ s$$^{-1}$$ is ______ K.
(Rounded-off to the nearest integer) [Given: The rate constant of the reaction is $$10^{-5}$$ s$$^{-1}$$ at 500 K.]
789
456
123
0.-
Clear All
NTA JEE Main 25th February 2021 Shift 1 - Chemistry - Question 60
Among the following, the number of halide(s) which is/are inert to hydrolysis is ______
(A) $$BF_3$$
(B) $$SiCl_4$$
(C) $$PCl_5$$
(D) $$SF_6$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




