NTA JEE Main 2nd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 2nd September 2020 Shift 2 - Question 31
Two elements A and B have similar chemical properties. They don't form solid hydrogencarbonates, but react with nitrogen to form nitrides. A and B, respectively, are:
NTA JEE Main 2nd September 2020 Shift 2 - Question 32
Arrange the following labelled hydrogens in decreasing order of acidity: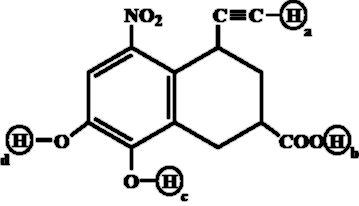
NTA JEE Main 2nd September 2020 Shift 2 - Question 33
An organic compound 'A' $$(C_9H_{10}O)$$ when treated with conc. HI undergoes cleavage to yield compound 'B' and 'C'. 'B' gives yellow precipitate with $$AgNO_3$$ where as 'C' tautomerizes to 'D'. 'D' gives positive iodoform test. 'A' could be:
NTA JEE Main 2nd September 2020 Shift 2 - Question 34
The major product of the following reaction is:
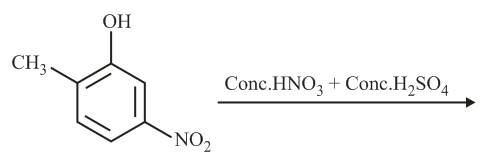
NTA JEE Main 2nd September 2020 Shift 2 - Question 35
The size of a raw mango shrinks to a much smaller size when kept in a concentrated salt solution. Which one of the following process can explain this?
NTA JEE Main 2nd September 2020 Shift 2 - Question 36
The results given in the below table were obtained during kinetic studies of the following reaction:
$$2A + B \to C + D$$
| Experiment | [A]/mol L$$^{-1}$$ | [B]/mol L$$^{-1}$$ | Initial rate/mol L$$^{-1}$$ min$$^{-1}$$ |
|---|---|---|---|
| I | 0.1 | 0.1 | $$6.00 \times 10^{-3}$$ |
| II | 0.1 | 0.2 | $$2.40 \times 10^{-2}$$ |
| III | 0.2 | 0.1 | $$1.20 \times 10^{-2}$$ |
| IV | X | 0.2 | $$7.20 \times 10^{-2}$$ |
| V | 0.3 | Y | $$2.88 \times 10^{-1}$$ |
X and Y in the given table are respectively:
NTA JEE Main 2nd September 2020 Shift 2 - Question 37
Amongst the following statements regarding adsorption, those that are valid are:
(a) $$\Delta H$$ becomes less negative as adsorption proceeds.
(b) On a given adsorbent, ammonia is adsorbed more than nitrogen gas.
(c) On adsorption, the residual force acting along the surface of the adsorbent increases
(d) With increase in temperature, the equilibrium concentration of adsorbate increases.
NTA JEE Main 2nd September 2020 Shift 2 - Question 38
Cast iron is used for the manufacture of:
NTA JEE Main 2nd September 2020 Shift 2 - Question 39
Simplified absorption spectra of three complexes ((i) and (ii) and (iii)) of $$M^{+n}$$ ion are provided below; their $$\lambda_{max}$$ values are marked as A, B and C respectively. The correct match between the complexes and their $$\lambda_{max}$$ values is: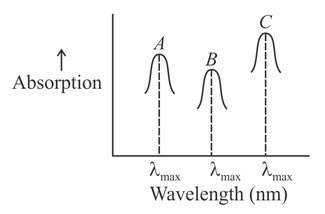
(i) $$[M(NCS)_6]^{(-6+n)}$$
(ii) $$[MF_6]^{(-6+n)}$$
(iii) $$[M(NH_3)_6]^{n+}$$
NTA JEE Main 2nd September 2020 Shift 2 - Question 40
The one that is not expected to show isomerism is:


