NTA JEE Main 2nd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 2nd September 2020 Shift 2 - Question 21
A square shaped hole of side $$l = \frac{a}{2}$$ is carved out at a distance $$d = \frac{a}{2}$$ from the centre 'O' of a uniform circular disk of radius a. If the distance of the centre of mass of the remaining portion from O is $$-\frac{a}{x}$$, value of X (to the nearest integer) is: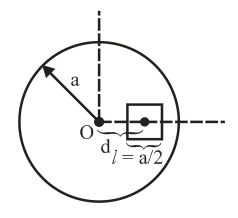
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 22
A particle of mass m is moving along the x-axis with initial velocity $$u\hat{i}$$. It collides elastically with a particle of mass 10m at rest and then moves with half its initial kinetic energy (see figure). If $$\sin\theta_1 = \sqrt{n}\sin\theta_2$$, then value of n is ___________.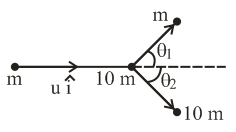
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 23
A wire of density $$9 \times 10^{-3}$$ kg cm$$^{-3}$$ is stretched between two clamps 1 m apart. The resulting strain in the wire is $$4.9 \times 10^{-4}$$. The lowest frequency of the transverse vibrations in the wire (Young's modulus of wire $$Y = 9 \times 10^{10}$$ Nm$$^{-2}$$), (to the nearest integer) is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 24
An ideal cell of emf 10V is connected in circuit shown in figure. Each resistance is $$2\Omega$$. The potential difference (in V) across the capacitor when it is fully charged is ___________.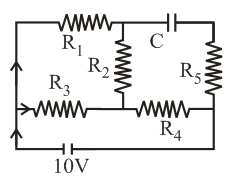
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 25
A light ray enters a solid glass sphere of refractive index $$\mu = \sqrt{3}$$ at an angle of incidence 60$$°$$. The ray is both reflected and refracted at the farther surface of the sphere. The angle (in degrees) between the reflected and refracted rays at this surface is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 26
The number of subshells associated with $$n = 4$$ and $$m = -2$$ quantum numbers is:
NTA JEE Main 2nd September 2020 Shift 2 - Question 27
Three elements X, Y and Z are in the 3$$^{rd}$$ period of the periodic table. The oxides of X, Y and Z, respectively, are basic, amphoteric and acidic. The correct order of the atomic numbers of X, Y and Z is:
NTA JEE Main 2nd September 2020 Shift 2 - Question 28
The shape/structure of $$[XeF_5]^-$$ and $$XeO_3F_2$$, respectively are:
NTA JEE Main 2nd September 2020 Shift 2 - Question 29
Match the type of interaction in column A with the distance dependence of their interaction energy in column B:
A B
(i) ion - ion (a) $$\frac{1}{r}$$
(ii) Dipole - dipole (b) $$\frac{1}{r^2}$$
(iii) London dispersion (c) $$\frac{1}{r^3}$$
(iv) $$\frac{1}{r^6}$$
NTA JEE Main 2nd September 2020 Shift 2 - Question 30
The molecular geometry of $$SF_6$$ is octahedral. What is the geometry of $$SF_4$$ (including lone pair(s) of electrons, if any)?


