NTA JEE Main 2nd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 2nd September 2020 Shift 2 - Question 41
The major product obtained from $$E_2$$-elimination of 3-bromo-2-fluoropentane is:
NTA JEE Main 2nd September 2020 Shift 2 - Question 42
Consider the reaction sequence given below:
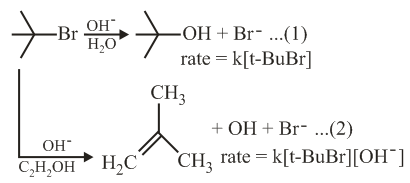
Which of the following statements is true:
NTA JEE Main 2nd September 2020 Shift 2 - Question 43
Two compounds A and B with same molecular formula $$(C_3H_6O)$$ undergo Grignard reaction with methylmagnesium bromide to give products C and D. Products C and D show following chemical tests:
| Test | C | D |
|---|---|---|
| Ceric ammonium nitrate Test | Positive | Positive |
| Lucas Test | Turbidity obtained after five minutes | Turbidity obtained Immediately |
| Iodoform Test | Positive | Negative |
C and D respectively are:
NTA JEE Main 2nd September 2020 Shift 2 - Question 44
If you spill a chemical toilet cleaning liquid on your hand, your first aid would be:
NTA JEE Main 2nd September 2020 Shift 2 - Question 45
The correct observation in the following reaction is:
$$Sucrose \xrightarrow[Cleavage \; (Hydrolysis)]{Glycosidic \; bond \; } A + B \xrightarrow[ Reagent]{Seliwanoff } ?$$
NTA JEE Main 2nd September 2020 Shift 2 - Question 46
The ratio of the mass percentages of 'C & H' and 'C & O' of a saturated acyclic organic compound 'X' are 4 : 1 and 3 : 4 respectively. Then, the moles of oxygen gas required for complete combustion of two moles of organic compound 'X' is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 47
The work function of sodium metal is $$4.41 \times 10^{-19}$$ J. If photons of wavelength 300 nm are incident on the metal, the kinetics energy of the ejected electrons will be $$(h = 6.63 \times 10^{-34}$$ J s; $$c = 3 \times 10^8$$ m s$$^{-1})$$ ___________ $$\times 10^{-21}$$ J.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 48
The heat of combustion of ethanol into carbon dioxides and water is $$-327$$ Kcal at constant pressure. The heat evolved (in cal) at constant volume at 27$$°$$C (if all gases behave ideally) is $$(R = 2$$ cal mol$$^{-1}$$ K$$^{-1})$$ ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 49
The oxidation states of transition metal atoms in $$K_2Cr_2O_7$$, $$KMnO_4$$ and $$K_2FeO_4$$, respectively, are x, y and z. The sum of x, y and z is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 2nd September 2020 Shift 2 - Question 50
For the disproportionation reaction $$2Cu^+(aq) \rightleftharpoons Cu(s) + Cu^{2+}(aq)$$ at 298K, ln K (where K is the equilibrium constant) is ___________ $$\times 10^{-1}$$.
Given: $$(E^°_{Cu^{2+}/Cu^+} = 0.16V, E^°_{Cu^+/Cu} = 0.52V, \frac{RT}{F} = 0.025)$$
789
456
123
0.-
Clear All


