NTA JEE Main 15th April 2018 Shift 2
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 2 - Question 31
For per gram of reactant, the maximum quantity of N$$_2$$ gas is produced in which of the following thermal decomposition reactions? (Given: Atomic wt. : Cr = 52u, Ba = 137u)
NTA JEE Main 15th April 2018 Shift 2 - Question 32
The de-Broglie's wavelength of electron present in first Bohr orbit of 'H' atom is:
NTA JEE Main 15th April 2018 Shift 2 - Question 33
The correct order of electron affinity is:
NTA JEE Main 15th April 2018 Shift 2 - Question 34
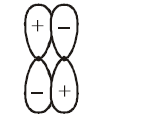
Which of the following best describes the diagram of molecular orbital?
NTA JEE Main 15th April 2018 Shift 2 - Question 35
$$\Delta_r G^\circ$$ at 500 K for substance 'S' in liquid state and gaseous state are +100.7 kcal mol$$^{-1}$$ and +103 kcal mol$$^{-1}$$, respectively. Vapour pressure of liquid 'S' at 500 K is approximately equal to: (R = 2 cal K$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 15th April 2018 Shift 2 - Question 36
Given (i) 2Fe$$_2$$O$$_3$$(s) $$\rightarrow$$ 4Fe(s) + 3O$$_2$$(g); $$\Delta_r G^\circ$$ = +1487.0 kJ mol$$^{-1}$$
(ii) 2CO(g) + O$$_2$$(g) $$\rightarrow$$ 2CO$$_2$$(g); $$\Delta_r G^\circ$$ = -514.4 kJ mol$$^{-1}$$
Free energy change, $$\Delta_r G^\circ$$ for the reaction 2Fe$$_2$$O$$_3$$(s) + 6CO(g) $$\rightarrow$$ 4Fe(s) + 6CO$$_2$$(g) will be:
NTA JEE Main 15th April 2018 Shift 2 - Question 37
At a certain temperature in a 5L vessel, 2 moles of carbon monoxide and 3 moles of chlorine were allowed to reach equilibrium according to the reaction, CO + Cl$$_2$$ $$\rightleftharpoons$$ COCl$$_2$$. At equilibrium, if one mole of CO is present then equilibrium constant (K$$_c$$) for the reaction is:
NTA JEE Main 15th April 2018 Shift 2 - Question 38
Following four solutions are prepared by mixing different volumes of NaOH and HCl of different concentrations, pH of which one of them will be equal to 1?
NTA JEE Main 15th April 2018 Shift 2 - Question 39
In KO$$_2$$, the nature of oxygen species and the oxidation state of oxygen atom are, respectively:
NTA JEE Main 15th April 2018 Shift 2 - Question 40
Lithium aluminium hydride reacts with silicon tetrachloride to form:


