NTA JEE Main 15th April 2018 Shift 2
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 2 - Question 41
The total number of optically active compounds formed in the following reaction is:
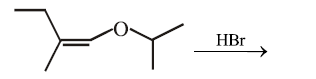
NTA JEE Main 15th April 2018 Shift 2 - Question 42
On the treatment of the following compound with a strong acid, the most susceptible site for bond cleavage is:
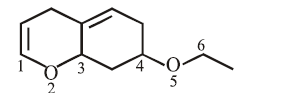
NTA JEE Main 15th April 2018 Shift 2 - Question 43
Two compounds I and II are eluted by column chromatography (adsorption of I > II). Which one of the following is a correct statement?
NTA JEE Main 15th April 2018 Shift 2 - Question 44
When 2-butyne is treated with H$$_2$$/Lindlar's catalyst, compound X is produced as the major product and when treated with Na/liq. NH$$_3$$ it produces Y as the major product. Which of the following statements is correct?
NTA JEE Main 15th April 2018 Shift 2 - Question 45
The major product formed in the following reaction is:
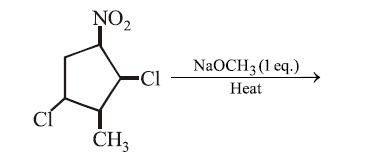
NTA JEE Main 15th April 2018 Shift 2 - Question 46
Biochemical oxygen demand (BOD) value can be a measure of water pollution caused by the organic matter. Which of the following statements is correct?
NTA JEE Main 15th April 2018 Shift 2 - Question 47
All of the following share the same crystal structure except:
NTA JEE Main 15th April 2018 Shift 2 - Question 48
Two 5 molal solutions are prepared by dissolving a non-electrolyte, non-volatile solute separately in the solvents X and Y. The molecular weights of the solvents are M$$_X$$ and M$$_Y$$, respectively where M$$_X$$ = $$\frac{3}{4}$$ M$$_Y$$. The relative lowering of vapour pressure of the solution in X is "m" times that of the solution in Y. Given that the number of moles of solute is very small in comparison to that of solvent, the value of "m" is:
NTA JEE Main 15th April 2018 Shift 2 - Question 49
For a first order reaction, A $$\rightarrow$$ P, t$$_{1/2}$$ (half-life) is 10 days. The time required for $$\frac{1}{4}$$th conversion of A (in days) is: (ln 2 = 0.693, ln 3 = 1.1)
NTA JEE Main 15th April 2018 Shift 2 - Question 50
If x gram of gas is adsorbed by m gram of adsorbent at pressure P the plot of log $$\frac{x}{m}$$ versus log P is linear. The slope of the plot is: (m and k are constants and n > 1)


