JEE Advanced 2024 Paper-2
For the following questions answer them individually
JEE Advanced 2024 Paper-2 - Question 41
The compound(s) having peroxide linkage is(are)
JEE Advanced 2024 Paper-2 - Question 42
To form a complete monolayer of acetic acid on 1 g of charcoal, 100 mL of 0.5 M acetic acid was used. Some of the acetic acid remained unadsorbed. To neutralize the unadsorbed acetic acid, 40 mL of 1 M NaOH solution was required. If each molecule of acetic acid occupies $$P \times 10^{-23}$$ $$m^2$$ surface area on charcoal, the value of P is ________.
[Use given data : Surface area of charcoal = $$1.5 \times 10^2$$ $$m^2 g^{-1}$$; Avogadro's number ($$N_A$$) = $$6.0 \times 10^{23}$$ $$mol^{-1}$$]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 43
Vessel-1 contains $$w_2$$ g of a non-volatile solute X dissolved in $$w_1$$ g of water. Vessel-2 contains $$w_2$$ g of another non-volatile solute Y dissolved in $$w_1$$ g of water. Both the vessels are at the same temperature and pressure. The molar mass of X is 80% of that of Y. The van't Hoff factor for X is 1.2 times of that of Y for their respective concentrations.
The elevation of boiling point for solution in Vessel-1 is ______ % of the solution in Vessel-2.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 44
For a double strand DNA, one strand is given below:
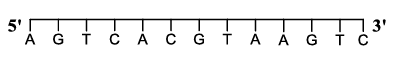
The amount of energy required to split the double strand DNA into two single strands is ______ kcal $$mol^{-1}$$.
[Given: Average energy per H-bond for A-T base pair = 1.0 kcal $$mol^{-1}$$, G-C base pair = 1.5 kcal $$mol^{-1}$$, and A-U base pair = 1.25 kcal $$mol^{-1}$$. Ignore electrostatic repulsion between the phosphate groups.]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 45
A sample initially contains only U-238 isotope of uranium. With time, some of the U-238 radioactively decays into Pb-206 while the rest of it remains undisintegrated.
When the age of the sample is $$P \times 10^8$$ years, the ratio of mass of Pb-206 to that of U-238 in the sample is found to be 7. The value of P is ______.
[Given : Half-life of U-238 is $$4.5 \times 10^9$$ years; $$\log_e 2$$ = 0.693]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 46
Among $$[Co(CN)_4]^{4-}$$, $$[Co(CO)_3(NO)]$$, $$XeF_4$$, $$[PCl_4]^+$$, $$[PdCl_4]^{2-}$$, $$[ICl_4]^-$$, $$[Cu(CN)_4]^{3-}$$ and $$P_4$$ the total number of species with tetrahedral geometry is ______.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 47
An organic compound P having molecular formula $$C_6H_6O_3$$ gives ferric chloride test and does not have intramolecular hydrogen bond. The compound P reacts with 3 equivalents of $$NH_2OH$$ to produce oxime Q. Treatment of P with excess methyl iodide in the presence of KOH produces compound R as the major product. Reaction of R with excess iso-butylmagnesium bromide followed by treatment with $$H_3O^+$$ gives compound S as the major product.
The total number of methyl ($$-CH_3$$) group(s) in compound S is ______.
789
456
123
0.-
Clear All
An organic compound P with molecular formula $$C_9H_{18}O_2$$ decolorizes bromine water and also shows positive iodoform test. P on ozonolysis followed by treatment with $$H_2O_2$$ gives Q and R. While compound Q shows positive iodoform test, compound R does not give positive iodoform test. Q and R on oxidation with pyridinium chlorochromate (PCC) followed by heating give S and T, respectively. Both S and T show positive iodoform test.
Complete copolymerization of 500 moles of Q and 500 moles of R gives one mole of a single acyclic copolymer U.
[Given, atomic mass: H = 1, C = 12, O = 16]
JEE Advanced 2024 Paper-2 - Question 48
Sum of number of oxygen atoms in S and T is ______.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 49
The molecular weight of U is ______.
789
456
123
0.-
Clear All
When potassium iodide is added to an aqueous solution of potassium ferricyanide, a reversible reaction is observed in which a complex P is formed. In a strong acidic medium, the equilibrium shifts completely towards P. Addition of zinc chloride to P in a slightly acidic medium results in a sparingly soluble complex Q.
JEE Advanced 2024 Paper-2 - Question 50
The number of moles of potassium iodide required to produce two moles of P is ______.
789
456
123
0.-
Clear All



