JEE Advanced 2024 Paper-2
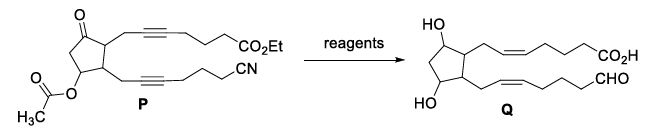
In a Young's double slit experiment, each of the two slits $$A$$ and $$B$$, as shown in the figure, are oscillating about their fixed center and with a mean separation of 0.8 mm. The distance between the slits at time $$t$$ is given by $$d = (0.8 + 0.04 \sin \omega t)$$ mm, where $$\omega = 0.08$$ rad s$$^{-1}$$. The distance of the screen from the slits is 1 m and the wavelength of the light used to illuminate the slits is 6000 Å. The interference pattern on the screen changes with time, while the central bright fringe (zeroth fringe) remains fixed at point $$O$$.
JEE Advanced 2024 Paper-2 - Question 31
The 8th bright fringe above the point $$O$$ oscillates with time between two extreme positions. The separation between these two extreme positions, in micrometer ($$\mu$$m), is ________.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 32
The maximum speed in $$\mu$$ m/s at which the $$8^{th}$$ bright fringe will move is ________.
789
456
123
0.-
Clear All
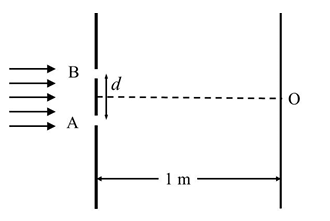
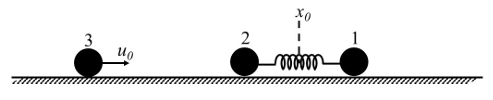
Two particles, 1 and 2, each of mass $$m$$, are connected by a massless spring, and are on a horizontal frictionless plane, as shown in the figure. Initially, the two particles, with their center of mass at $$x_0$$, are oscillating with amplitude $$a$$ and angular frequency $$\omega$$. Thus, their positions at time $$t$$ are given by $$x_1(t) = (x_0 + d) + a \sin \omega t$$ and $$x_2(t) = (x_0 - d) - a \sin \omega t$$, respectively, where $$d > 2a$$. Particle 3 of mass $$m$$ moves towards this system with speed $$u_0 = a\omega/2$$, and undergoes instantaneous elastic collision with particle 2, at time $$t_0$$. Finally, particles 1 and 2 acquire a center of mass speed $$v_{cm}$$ and oscillate with amplitude $$b$$ and the same angular frequency $$\omega$$.
JEE Advanced 2024 Paper-2 - Question 33
If the collision occurs at time $$t_0 = 0$$, the value of $$v_{cm}/(a\omega)$$ will be ________.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-2 - Question 34
If the collision occurs at time $$t_0 = \pi/(2\omega)$$, then the value of $$4b^2/a^2$$ will be ________.
789
456
123
0.-
Clear All
For the following questions answer them individually
JEE Advanced 2024 Paper-2 - Question 35
According to Bohr's model, the highest kinetic energy is associated with the electron in the
JEE Advanced 2024 Paper-2 - Question 36
In a metal deficient oxide sample, $$M_XY_2O_4$$ (M and Y are metals), M is present in both +2 and +3 oxidation states and Y is in +3 oxidation state. If the fraction of $$M^{2+}$$ ions present in M is $$\frac{1}{3}$$, the value of X is ______.
JEE Advanced 2024 Paper-2 - Question 37
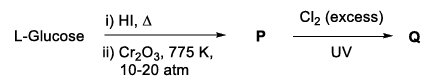
In the following reaction sequence, the major product Q is
JEE Advanced 2024 Paper-2 - Question 38
The species formed on fluorination of phosphorus pentachloride in a polar organic solvent are
JEE Advanced 2024 Paper-2 - Question 39
An aqueous solution of hydrazine ($$N_2H_4$$) is electrochemically oxidized by $$O_2$$, thereby releasing chemical energy in the form of electrical energy. One of the products generated from the electrochemical reaction is $$N_2(g)$$.
Choose the correct statement(s) about the above process
JEE Advanced 2024 Paper-2 - Question 40
The option(s) with correct sequence of reagents for the conversion of P to Q is(are)