JEE Advanced 2024 Paper-1
For the following questions answer them individually
JEE Advanced 2024 Paper-1 - Question 41
The option(s) in which at least three molecules follow Octet Rule is(are)
JEE Advanced 2024 Paper-1 - Question 42
Consider the following volume-temperature (V-T) diagram for the expansion of 5 moles of an ideal monoatomic gas.
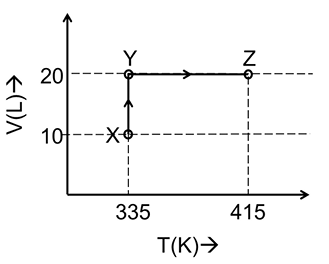
Considering only P-V work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $$X \rightarrow Y \rightarrow Z$$ is ______.
[Use the given data: Molar heat capacity of the gas for the given temperature range, $$C_{V,m} = 12$$ J $$K^{-1}$$ $$mol^{-1}$$ and gas constant, $$R = 8.3$$ J $$K^{-1}$$ $$mol^{-1}$$]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 43
Consider the following reaction,
$$2H_2(g) + 2NO(g) \rightarrow N_2(g) + 2H_2O(g)$$
which follows the mechanism given below:
$$2NO(g) \underset{k_{-1}}{\overset{k_1}{\rightleftharpoons}} N_2O_2(g)$$ (fast equilibrium)
$$N_2O_2(g) + H_2(g) \overset{k_2}{\rightarrow} N_2O(g) + H_2O(g)$$ (slow reaction)
$$N_2O(g) + H_2(g) \overset{k_3}{\rightarrow} N_2(g) + H_2O(g)$$ (fast reaction)
The order of the reaction is ______?
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 44
Complete reaction of acetaldehyde with excess formaldehyde, upon heating with conc. NaOH solution, gives P and Q. Compound P does not give Tollens' test, whereas Q on acidification gives positive Tollens' test. Treatment of P with excess cyclohexanone in the presence of catalytic amount of p-toluenesulfonic acid (PTSA) gives product R.
Sum of the number of methylene groups ($$-CH_2-$$) and oxygen atoms in R is ______.
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 45
Among $$V(CO)_6$$, $$Cr(CO)_5$$, $$Cu(CO)_3$$, $$Mn(CO)_5$$, $$Fe(CO)_5$$, $$[Co(CO)_3]^{3-}$$, $$[Cr(CO)_4]^{4-}$$, and $$Ir(CO)_3$$, the total number of species isoelectronic with $$Ni(CO)_4$$ is ______.
[Given atomic number : V = 23, Cr = 24, Mn = 25, Fe = 26, Co = 27, Ni = 28, Cu = 29, Ir = 77]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 46
In the following reaction sequence, the major product P is formed.
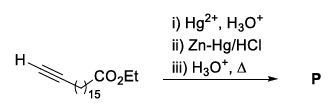
Glycerol reacts completely with excess P in the presence of an acid catalyst to form Q. Reaction of Q with excess NaOH followed by the treatment with $$CaCl_2$$ yields Ca-soap R, quantitatively. Starting with one mole of Q, the amount of R produced in gram is ______.
[Given, atomic weight: H = 1, C = 12, N = 14, O = 16, Na = 23, Cl = 35, Ca = 40]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 47
Among the following complexes, the total number of diamagnetic species is ______.
$$[Mn(NH_3)_6]^{3+}$$, $$[MnCl_6]^{3-}$$, $$[FeF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Fe(NH_3)_6]^{3+}$$ and $$[Co(en)_3]^{3+}$$
[Given, atomic number: Mn = 25, Fe = 26, Co = 27; en = $$H_2NCH_2CH_2NH_2$$]
789
456
123
0.-
Clear All
JEE Advanced 2024 Paper-1 - Question 48
In a conductometric titration, small volume of titrant of higher concentration is added stepwise to a larger volume of titrate of much lower concentration, and the conductance is measured after each addition.
The limiting ionic conductivity ($$\Lambda_0$$) values (in mS $$m^2$$ $$mol^{-1}$$) for different ions in aqueous solutions are given below:
| Ions | $$Ag^+$$ | $$K^+$$ | $$Na^+$$ | $$H^+$$ | $$NO_3^-$$ | $$Cl^-$$ | $$SO_4^{2-}$$ | $$OH^-$$ | $$CH_3COO^-$$ |
|---|---|---|---|---|---|---|---|---|---|
| $$\Lambda_0$$ | 6.2 | 7.4 | 5.0 | 35.0 | 7.2 | 7.6 | 16.0 | 19.9 | 4.1 |
For different combinations of titrates and titrants given in List-I, the graphs of 'conductance' versus 'volume of titrant' are given in List-II.
Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | Titrate: KCl Titrant: $$AgNO_3$$ | (1) | 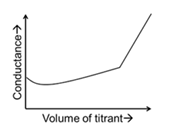 |
| (Q) | Titrate: $$AgNO_3$$ Titrant: KCl | (2) | 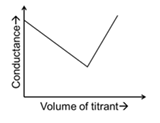 |
| (R) | Titrate: NaOH Titrant: HCl | (3) | 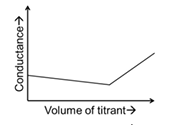 |
| (S) | Titrate: NaOH Titrant: $$CH_3COOH$$ | (4) | 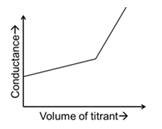 |
| (5) | 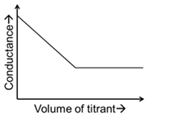 | ||
JEE Advanced 2024 Paper-1 - Question 49
Based on VSEPR model, match the xenon compounds given in List-I with the corresponding geometries and the number of lone pairs on xenon given in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | $$XeF_2$$ | (1) | Trigonal bipyramidal and two lone pair of electrons |
| (Q) | $$XeF_4$$ | (2) | Tetrahedral and one lone pair of electrons |
| (R) | $$XeO_3$$ | (3) | Octahedral and two lone pair of electrons |
| (S) | $$XeO_3F_2$$ | (4) | Trigonal bipyramidal and no lone pair of electrons |
| (5) | Trigonal bipyramidal and three lone pair of electrons | ||
JEE Advanced 2024 Paper-1 - Question 50
List-I contains various reaction sequences and List-II contains the possible products. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | 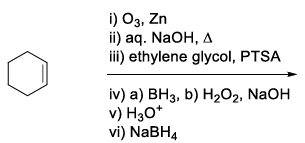 | (1) | 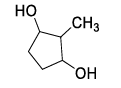 |
| (Q) | 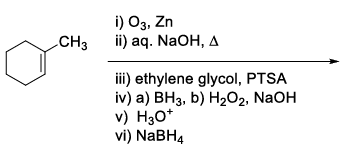 | (2) | 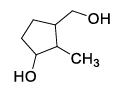 |
| (R) | 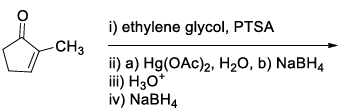 | (3) | 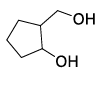 |
| (S) | 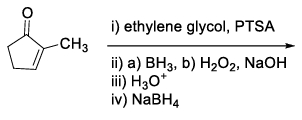 | (4) | 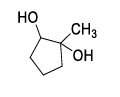 |
| (5) | 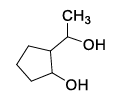 | ||


