JEE Advanced 2023 Paper-1
For the following questions answer them individually
JEE Advanced 2023 Paper-1 - Question 41
In the given reaction scheme, P is a phenyl alkyl ether, Q is an aromatic compound; R and S are the major products.
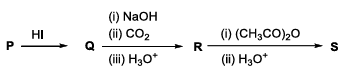
The correct statement about S is
JEE Advanced 2023 Paper-1 - Question 42
The stoichiometric reaction of 516 g of dimethyldichlorosilane with water results in a tetrameric cyclic product X in 75% yield. The weight (in g) of X obtained is ____.
[Use, molar mass (g mol$$^{-1}$$): H = 1, C = 12, O = 16, Si = 28, Cl = 35.5]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 43
A gas has a compressibility factor of 0.5 and a molar volume of 0.4 dm$$^3$$ mol$$^{-1}$$ at a temperature of 800 K and pressure x atm. If it shows ideal gas behaviour at the same temperature and pressure, the molar volume will be y dm$$^3$$ mol$$^{-1}$$. The value of x/y is ____.
[Use: Gas constant, R = $$8 \times 10^{-2}$$ L atm K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 44
The plot of log $$k_f$$ versus $$1/T$$ for a reversible reaction A(g) $$\rightleftharpoons$$ P(g) is shown.
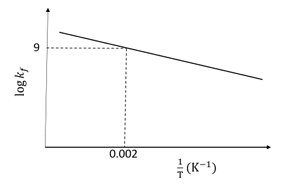
Pre-exponential factors for the forward and backward reactions are $$10^{15}$$ s$$^{-1}$$ and $$10^{11}$$ s$$^{-1}$$, respectively. If the value of log K for the reaction at 500 K is 6, the value of $$|\log k_b|$$ at 250 K is ______.
[K = equilibrium constant of the reaction,
$$k_f$$ = rate constant of forward reaction,
$$k_b$$ = rate constant of backward reaction]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 45
One mole of an ideal monoatomic gas undergoes two reversible processes (A $$\to$$ B and B $$\to$$ C) as shown in the given figure:
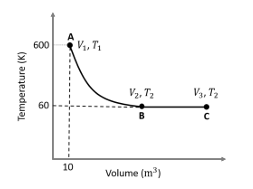
A $$\to$$ B is an adiabatic process. If the total heat absorbed in the entire process (A $$\to$$ B and B $$\to$$ C) is $$RT_2 \ln 10$$, the value of $$2 \log V_3$$ is ________.
[Use, molar heat capacity of the gas at constant pressure, $$C_{p,m} = \frac{5}{2}R$$]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 46
In a one-litre flask, 6 moles of A undergoes the reaction A(g) $$\rightleftharpoons$$ P(g). The progress of product formation at two temperatures (in Kelvin), $$T_1$$ and $$T_2$$, is shown in the figure:
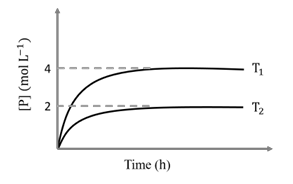
If $$T_1 = 2T_2$$ and $$(\Delta G_2^\circ - \Delta G_1^\circ) = RT_2 \ln x$$, then the value of x is ______.
[$$\Delta G_1^\circ$$ and $$\Delta G_2^\circ$$ are standard Gibb's free energy change for the reaction at temperatures $$T_1$$ and $$T_2$$, respectively.]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 47
The total number of sp$$^2$$ hybridised carbon atoms in the major product P (a non-heterocyclic compound) of the following reaction is ______.
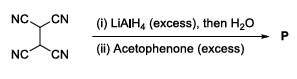
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-1 - Question 48
Match the reactions (in the given stoichiometry of the reactants) in List-I with one of their products given in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | $$P_2O_3 + 3H_2O \to$$ | (1) | $$P(O)(OCH_3)Cl_2$$ |
| (Q) | $$P_4 + 3NaOH + 3H_2O \to$$ | (2) | $$H_3PO_3$$ |
| (R) | $$PCl_5 + CH_3COOH \to$$ | (3) | $$PH_3$$ |
| (S) | $$H_3PO_2 + 2H_2O + 4AgNO_3 \to$$ | (4) | $$POCl_3$$ |
| (5) | $$H_3PO_4$$ | ||
JEE Advanced 2023 Paper-1 - Question 49
Match the electronic configurations in List-I with appropriate metal complex ions in List-II and choose the correct option.
[Atomic Number: Fe = 26, Mn = 25, Co = 27]
| List-I | List-II | ||
|---|---|---|---|
| (P) | $$t_{2g}^6 e_g^0$$ | (1) | $$[Fe(H_2O)_6]^{2+}$$ |
| (Q) | $$t_{2g}^3 e_g^2$$ | (2) | $$[Mn(H_2O)_6]^{2+}$$ |
| (R) | $$e^2 t_2^3$$ | (3) | $$[Co(NH_3)_6]^{3+}$$ |
| (S) | $$t_{2g}^4 e_g^2$$ | (4) | $$[FeCl_4]^{-}$$ |
| (5) | $$[CoCl_4]^{2-}$$ | ||
JEE Advanced 2023 Paper-1 - Question 50
Match the reactions in List-I with the features of their products in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | (-)-1-Bromo-2-ethylpentane (single enantiomer) $$\xrightarrow[S_N2 \text{ reaction}]{\text{aq. NaOH}}$$ | (1) | Inversion of configuration |
| (Q) | (-)-2-Bromopentane (single enantiomer) $$\xrightarrow[S_N2 \text{ reaction}]{\text{aq. NaOH}}$$ | (2) | Retention of configuration |
| (R) | (-)-3-Bromo-3-methylhexane (single enantiomer) $$\xrightarrow[S_N1 \text{ reaction}]{\text{aq. NaOH}}$$ | (3) | Mixture of enantiomers |
| (S) | 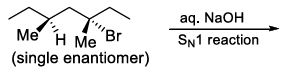 | (4) | Mixture of structural isomers |
| (5) | Mixture of diastereomers | ||



