Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following electrochemical cell at standard condition.
$$Au(s)|QH_2,Q|NH_4X(0.01M)||Ag^+(1M)|Ag(s)$$
$$E_{cell} = +0.4V$$
The couple $$QH_2/Q$$ represents quinhydrone electrode, the half cell reaction is given below:
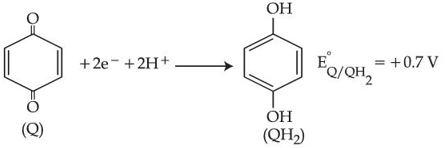
[Given: $$E^o_{Ag^+/Ag} = +0.8V$$ and $$\frac{2.303RT}{F} = 0.06V$$]
The $$pK_b$$ value of the ammonium halide salt ($$NH_4X$$) used here is ______. (nearest integer)
Correct Answer: 6
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
