Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
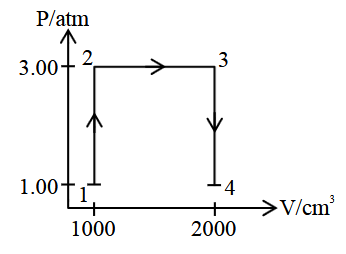
A perfect gas (0.1 mol) having $$\bar{C}_v = 1.50 R$$ (independent of temperature) undergoes the transformation from point 1 to point 4 as shown in the P-V diagram. If each step is reversible, the total work done (w) while going from point 1 to point 4 is $$(-) \text{____}$$ J (nearest integer).
[Given: $$R = 0.082$$ L atm $$K^{-1} mol^{-1}$$]
Correct Answer: 304
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.