Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Resonance in $$X_2Y$$ can be represented as
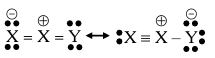
The enthalpy of formation of X_2Y\left( X\equiv X(g)+\frac{1}{2}Y=Y(g)\rightarrow X_2 Y(g)\right) is 80 kJ mol$$^{-1}$$. The magnitude of resonance energy of $$X_2Y$$ is _____ kJ mol$$^{-1}$$ (nearest integer value). Given: Bond energies of $$X \equiv X$$, $$X = X$$, $$Y = Y$$ and $$X = Y$$ are 940, 410, 500 and 602 kJ mol$$^{-1}$$ respectively.
valence X : 3, Y : 2
Correct Answer: 98
The resonance energy of a molecule is the extra stabilisation it enjoys due to resonance.
Mathematically,
$$\text{Resonance energy}= \left( \Delta H_f^{\,\text{calculated (one structure)}} \right) - \left( \Delta H_f^{\,\text{experimental}} \right)$$
The question gives the experimental enthalpy of formation of $$X_2Y$$ as
$$\Delta H_f^{\,\text{exp}} = 80\ \text{kJ mol}^{-1}$$
To obtain $$\Delta H_f^{\,\text{calculated}}$$ we choose any one canonical (non-resonating) structure.
A convenient choice for $$X_2Y$$ is
$$X=X - Y\;,$$
i.e. one $$X=X$$ double bond and one $$X=Y$$ double bond.
The enthalpy of formation is obtained from the usual bond-energy relation
$$\Delta H_f = \sum \text{(bond energies of bonds broken)} \;-\; \sum \text{(bond energies of bonds formed)}$$
Step 1: Bonds broken (to obtain free atoms)
We start from the elemental forms:
• 1 mol $$X_2$$ contains one $$X\equiv X$$ triple bond
Energy to break it = $$E_{X\equiv X}=940\ \text{kJ}$$
• ½ mol $$Y_2$$ contains half a $$Y=Y$$ double bond
Energy to break it = $$\tfrac12\,E_{Y=Y}
=\tfrac12 \times 500
=250\ \text{kJ}$$
Total energy required to break reactant bonds:
$$E_{\text{broken}} = 940 + 250 = 1190\ \text{kJ}$$
Step 2: Bonds formed in the chosen structure of $$X_2Y$$
• One $$X=X$$ double bond energy released = $$E_{X=X}=410\ \text{kJ}$$
• One $$X=Y$$ double bond energy released = $$E_{X=Y}=602\ \text{kJ}$$
Total energy released on forming product bonds:
$$E_{\text{formed}} = 410 + 602 = 1012\ \text{kJ}$$
Step 3: Calculated enthalpy of formation (one structure)
$$\Delta H_f^{\,\text{calc}} = E_{\text{broken}} - E_{\text{formed}} = 1190 - 1012 = 178\ \text{kJ mol}^{-1}$$
Step 4: Resonance energy
$$\text{Resonance energy} = \Delta H_f^{\,\text{calc}} - \Delta H_f^{\,\text{exp}} = 178 - 80 = 98\ \text{kJ mol}^{-1}$$
Hence, the magnitude of the resonance energy of $$X_2Y$$ is 98 kJ mol$$^{-1}$$.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
