Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
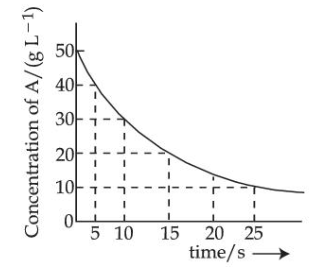
For the reaction $$A \to B$$ the following graph was obtained. The time required (in seconds) for the concentration of A to reduce to 2.5 g L$$^{-1}$$ (if the initial concentration of A was 50 g L$$^{-1}$$) is _________.
(Nearest integer)
Given : $$\log 2 = 0.3010$$
Correct Answer: 43
Create a FREE account and get:
Educational materials for JEE preparation
