Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
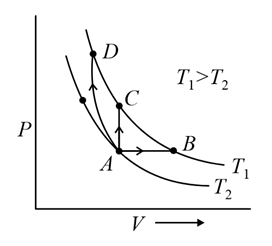
Three different processes that can occur in an ideal monoatomic gas are shown in the $$P$$ vs $$V$$ diagram. The paths are labelled as $$A \to B$$, $$A \to C$$ and $$A \to D$$. The change in internal energies during these processes are taken as $$E_{AB}$$, $$E_{AC}$$ and $$E_{AD}$$ and the work done as $$W_{AB}$$, $$W_{AC}$$ and $$W_{AD}$$. The correct relation between these parameters are:
We are dealing with an ideal mono-atomic gas. For such a gas the internal energy is a pure function of temperature alone and is given by the well-known expression
$$U=\tfrac32\,nRT.$$
Consequently, the change in internal energy between any two states depends only on the initial and final temperatures:
$$\Delta U=\tfrac32\,nR\,(T_{\text{final}}-T_{\text{initial}}).$$
In the $$P\!-\!V$$ diagram all three processes start from the common state $$A(P_A,V_A,T_A)$$ and terminate at the three distinct points $$B,\;C,\;D$$. A careful inspection of the diagram shows that the points $$B,\;C,\;D$$ lie on the same isothermal curve that passes through none of the other points. Algebraically that statement reads
$$P_BV_B=P_CV_C=P_DV_D=k\quad(\text{constant}).$$
Using the ideal-gas equation $$PV=nRT$$ this instantly gives
$$T_B=T_C=T_D=T',$$
where $$T'$$ is some temperature (which may or may not equal $$T_A$$). Because all three final temperatures are equal, the change in internal energy from the common initial state $$A$$ to any of the three final states is identical:
$$E_{AB}=\Delta U_{AB}=\tfrac32\,nR\,(T'-T_A),$$
$$E_{AC}=\Delta U_{AC}=\tfrac32\,nR\,(T'-T_A),$$
$$E_{AD}=\Delta U_{AD}=\tfrac32\,nR\,(T'-T_A).$$
Hence
$$E_{AB}=E_{AC}=E_{AD}.$$
Next we examine the work done, remembering the definition
$$W=\int_{V_{\text{initial}}}^{V_{\text{final}}}P\,dV.$$
Sign conventions to keep in mind:
• When the gas expands, $$dV>0$$ so $$W>0$$(positive work done by the gas).
• When the gas undergoes an isochoric (constant-volume) change, $$dV=0$$ so $$W=0$$(no work).
• When the gas is compressed, $$dV<0$$ so $$W<0$$(negative work, work done on the gas).
Looking again at the geometry of the three paths:
• Along $$A\to B$$ the curve clearly moves to a larger volume: $$V_B>V_A$$. Therefore $$W_{AB}>0.$
• Along $$A\to C$$ the path is vertical, i.e. the volume is unchanged: $$V_C=V_A$$. Hence $$W_{AC}=0.$
• Along $$A\to D$$ the path proceeds to a smaller volume: $$V_D<V_A$$. Consequently $$W_{AD}<0.$
Collecting all the deductions we have
$$E_{AB}=E_{AC}=E_{AD},\qquad W_{AB}>0,\qquad W_{AC}=0,\qquad W_{AD}<0.$$
This set of equalities and inequalities is exactly the one listed in Option B of the question.
Hence, the correct answer is Option B.
Create a FREE account and get:
Educational materials for JEE preparation
