Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
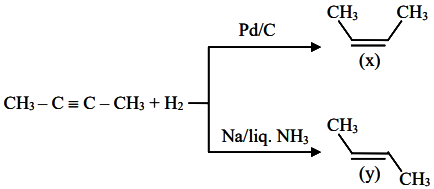
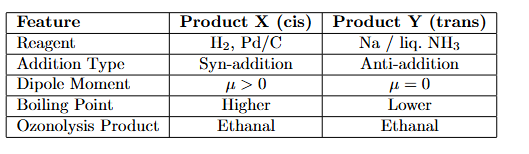
But-2-yne and hydrogen (one mole each) are separately treated with (i) Pd/C and (ii) Na/ liq. NH3 to give the products X and Y respectively.
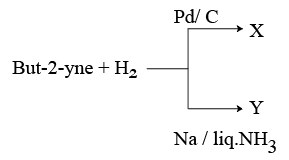
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of X is zero.
C. Boiling point of X is higher than Y.
D. X and Y react with $$O_{3}/Zn+H_{2}O$$ to give different products.
Choose the correct answer from the options given below :
Create a FREE account and get:
Educational materials for JEE preparation
