Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
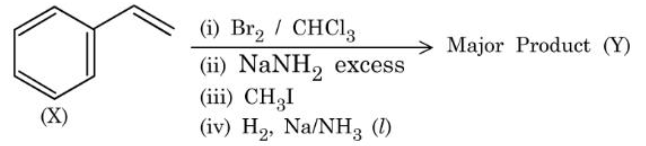
Compound (X) is subjected to the sequence of reactions as shown above:
Molar mass of the major product (Y) formed is ______ g mol$$^{-1}$$.
(Given molar mass in g mol$$^{-1}$$ C:12, H: 1, O: 16)
Step-1 Identification of the starting alkyne X
The product Y finally obtained after the dissolving-metal reduction (Na/NH3(l) + H2) must be a trans (E)-alkene because $$\text{Na}/\text{NH}_3(l)$$ converts an internal alkyne into a trans-alkene without disturbing an aromatic ring if present.
A molar mass of 118 g mol-1 corresponds to the molecular formula $$C_9H_{10}$$ ( $$9\times 12 + 10\times 1 = 118$$ ). Such a formula suggests the presence of a benzene ring ( $$C_6H_5$$ ) attached to a three-carbon side chain ( $$C_3H_5$$ ).
Therefore, compound Y is most plausibly trans-1-phenyl-1-propene (trans-β-methyl-styrene): $$\text{Ph-CH}=CH-CH_3$$.
Step-2 Retracing the sequence from Y to X
(i) The immediate precursor of this alkene in the given sequence must be the corresponding internal alkyne, $$\text{Ph-C}\equiv\text{C-CH}_3$$, because dissolving-metal reduction adds two hydrogens across a C≡C bond to give the trans-C=C product.
(ii) This alkyne is obtained by alkylating the sodium acetylide of a terminal alkyne with $$\text{CH}_3\text{I}$$. Hence the terminal alkyne just before step (iii) is phenylacetylene, $$\text{Ph-C}\equiv\text{C-H}$$, whose acidic proton has been removed by excess $$\text{NaNH}_2$$ to give $$\text{Ph-C}\equiv\text{C}^-\,\text{Na}^+$$.
(iii) That sodium acetylide itself is produced from the vic-dibromoalkene obtained in step (i) by double dehydro-bromination with excess $$\text{NaNH}_2$$.
(iv) Finally, the vic-dibromoalkene arises from the addition of one equivalent of $$\text{Br}_2$$ to the starting alkyne X.
Thus the only alkyne that fits all the steps is phenylacetylene, $$\text{Ph-C}\equiv\text{C-H}$$.
Step-3 Verification of the whole sequence
Case 1: $$\text{Ph-C}\equiv\text{C-H}\xrightarrow{\text{Br}_2/\text{CHCl}_3}$$ trans-1,2-dibromostyrene.
Case 2: trans-1,2-dibromostyrene $$\xrightarrow[\text{excess}]{\text{NaNH}_2}$$ phenylacetylene (regenerated) and then $$\text{Ph-C}\equiv\text{C}^-\,\text{Na}^+$$.
Case 3: $$\text{Ph-C}\equiv\text{C}^-\,\text{Na}^+\xrightarrow{\text{CH}_3\text{I}}$$ 1-phenyl-1-butyne, $$\text{Ph-C}\equiv\text{C-CH}_3$$.
Case 4: 1-phenyl-1-butyne $$\xrightarrow{\text{H}_2,\ \text{Na}/\text{NH}_3(l)}$$ trans-1-phenyl-1-propene (compound Y), $$\text{Ph-CH}=CH-CH_3$$.
Step-4 Molar-mass calculation of Y
Number of atoms in Y: 9 C and 10 H.
Molar mass $$M = 9\times 12\;(\text{C}) + 10\times 1\;(\text{H}) = 108 + 10 = 118\ \text{g mol}^{-1}$$.
Hence, the molar mass of compound Y is 118 g mol-1.
Option B which is: 118
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
