Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
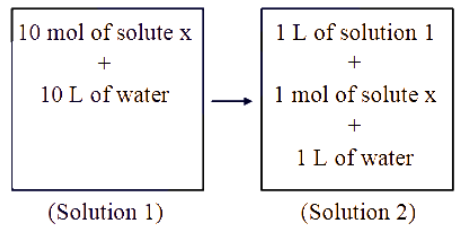
Which of the following properties will change when system containing solution 1 will become solution 2 ?
First, list the given data for the two systems.
Solution 1 (stock solution): 10 mol solute x + 10 L water.
Hence, the concentration is $$\frac{10\;\text{mol}}{10\;\text{L}} = 1\;\text{M}$$.
Solution 2 is prepared by mixing 1 L of Solution 1 with 1 mol of solute x and 1 L of water.
• Solute coming from 1 L of Solution 1 = 1 mol.
• Extra solute added = 1 mol.
Total solute in Solution 2 = 1 mol + 1 mol = 2 mol.
• Volume coming from 1 L of Solution 1 = 1 L.
• Extra water added = 1 L.
Total volume of Solution 2 = 1 L + 1 L = 2 L.
Therefore, the concentration of Solution 2 is $$\frac{2\;\text{mol}}{2\;\text{L}} = 1\;\text{M}$$, exactly the same as Solution 1.
Now analyse each property.
Case A: Molar heat capacity (heat capacity per mole of solution) is an intensive property; it depends only on composition, not on the amount. Because the mole-ratio of solute to solvent remains 1 M for both solutions, the molar heat capacity stays unchanged.
Case B: Density is also an intensive property (mass per unit volume). Since the composition is identical in both solutions, the density remains the same.
Case C: Concentration has already been shown to stay at 1 M. Hence, no change occurs.
Case D: Gibbs free energy $$G$$ is an extensive property; it is directly proportional to the amount of matter present. Solution 2 contains twice the amount of both solute and solvent compared with the 1 L portion of Solution 1, so its total Gibbs free energy is different (specifically, doubled) from that of Solution 1.
Therefore, the only property that changes when the system passes from Solution 1 to Solution 2 is the Gibbs free energy.
Correct choice: Option D (Gibbs free energy).
Create a FREE account and get:
Educational materials for JEE preparation
