Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
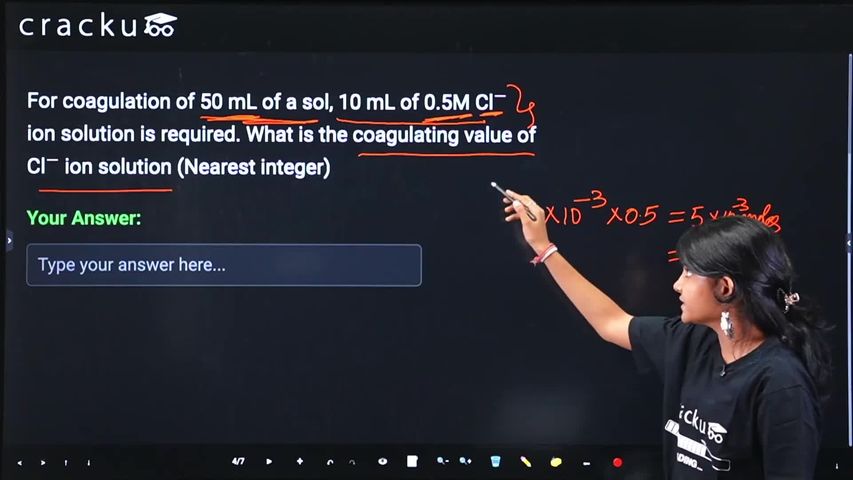
For coagulation of 50 mL of a sol, 10 mL of 0.5M Cl$$^-$$ ion solution is required. What is the coagulating value of Cl$$^-$$ ion solution (Nearest integer)
Correct Answer: 100
The coagulating value is defined as the number of millimoles of electrolyte required to coagulate 1 liter of a sol.
Given:
First, calculate the millimoles of Cl⁻ ions used:
Concentration = 0.5 M = 0.5 mol/L
Volume of Cl⁻ solution = 10 mL = 0.01 L
Moles of Cl⁻ = concentration × volume = $$0.5 \times 0.01 = 0.005$$ mol
Convert to millimoles: $$0.005 \times 1000 = 5$$ millimoles
Thus, 5 millimoles of Cl⁻ ions coagulate 50 mL of sol.
Now, find the millimoles required to coagulate 1 liter (1000 mL) of sol:
Using proportion:
$$\frac{5 \text{ millimoles}}{50 \text{ mL}} = \frac{x \text{ millimoles}}{1000 \text{ mL}}$$
Solve for $$x$$:
$$x = \frac{5 \times 1000}{50} = \frac{5000}{50} = 100$$ millimoles
Therefore, the coagulating value is 100 millimoles per liter.
The nearest integer is 100.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.