Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements :
$$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$ and 1 cal = 4.2 J
Statement I : When $$E_a = 12.6$$ kcal/mol, the room temperature rate constant is doubled by a 10 $$^\circ$$C increase in temperature (298 K to 308 K)
Statement II : For a first order reactions $$A \to B$$,
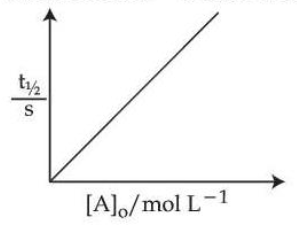
Here $$[A]_0$$ is the initial concentration of A and $$t_{1/2}$$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
