Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the List I with List II:
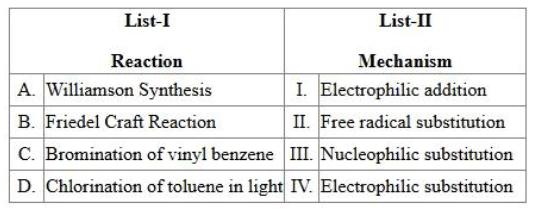
Choose the correct answer from the options given below
For each reaction we first identify the active intermediate and the bond-making/breaking pattern, then relate it to the standard mechanism types listed.
Case A: Williamson synthesis
The process is
$$R\!-\!O^- + R'X \rightarrow R\!-\!O\!-\!R' + X^-$$.
An alkoxide ion (strong nucleophile) attacks an alkyl halide in a one-step $$\text{S}\!_\text{N}2$$ fashion. A nucleophile substitutes for the leaving halide ion, so the mechanism is $$\textbf{Nucleophilic substitution (III)}$$.
Case B: Friedel-Crafts reaction
Example: $$C_6H_6 + RCl + AlCl_3 \rightarrow C_6H_5R + HCl$$.
The Lewis acid $$AlCl_3$$ generates the electrophile $$R^+$$ (or $$Ac^+$$ in acylation). This electrophile then substitutes a hydrogen on the aromatic ring via the classical arenium ion pathway. Therefore the mechanism is $$\textbf{Electrophilic substitution (IV)}$$.
Case C: Bromination of vinyl benzene (styrene)
Styrene is an alkene; bromine adds across its double bond:
$$C_6H_5CH{=}CH_2 + Br_2 \rightarrow C_6H_5CHBr\!-\!CH_2Br$$.
The $$Br_2$$ molecule polarises to give an electrophilic $$Br^+$$ that attacks the $$\pi$$-bond, followed by capture of $$Br^-$$. Addition of an electrophile to a double bond is classified as $$\textbf{Electrophilic addition (I)}$$.
Case D: Chlorination of toluene in sunlight
Under UV light, $$Cl_2$$ homolytically cleaves to give chlorine radicals. Chain propagation at the benzylic C-H bond produces benzyl chloride:
$$C_6H_5CH_3 + Cl_2 \xrightarrow{h\nu} C_6H_5CH_2Cl + HCl$$.
This is a radical chain process, so the mechanism is $$\textbf{Free radical substitution (II)}$$.
Collecting the matches:
A → III, B → IV, C → I, D → II
The option containing this set is Option C $$\longrightarrow\ $$A-III, B-IV, C-I, D-II.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
