Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the graph of Gibbs free energy G vs extent of reaction. The number of statement/s from the following which are true with respect to points (a), (b) and (c) is ______
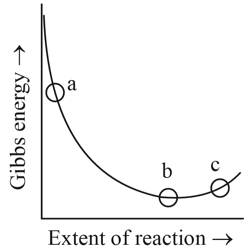
A. Reaction is spontaneous at (a) and (b)
B. Reaction is at equilibrium at point (b) and non-spontaneous at point (c)
C. Reaction is spontaneous at (a) and non-spontaneous at (c)
D. Reaction is non-spontaneous at (a) and (b)
Correct Answer: 2
Create a FREE account and get:
Educational materials for JEE preparation
