Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
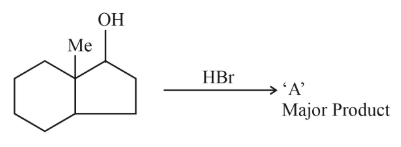
The number of hyperconjugation structures involved to stabilize carbocation formed in the reaction of 2-methylcyclohexan-1-ol with HBr is _______
Correct Answer: 7
When 2-methylcyclohexan-1-ol reacts with HBr, the OH group is first protonated to form a good leaving group ($$H_2O$$), which departs to give a secondary carbocation at C1.
Now, this secondary carbocation at C1 undergoes a 1,2-hydride shift from C2 to form a more stable tertiary carbocation at C2 (the carbon bearing the methyl group).
We have the tertiary carbocation at C2 bonded to:
- C1 (which now has 2 hydrogen atoms since OH left as water)
- C3 (which has 2 hydrogen atoms)
- $$CH_3$$ group (which has 3 hydrogen atoms)
Each $$\alpha$$-hydrogen can participate in hyperconjugation with the empty p-orbital of the carbocation.
So, total number of hyperconjugating hydrogens = 2 + 2 + 3 = 7.
Hence, the number of hyperconjugation structures is $$7$$.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.