Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
List-I contains various reaction sequences and List-II contains the possible products. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) |  | (1) |  |
| (Q) |  | (2) |  |
| (R) | 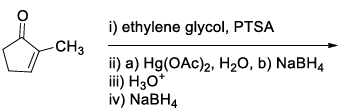 | (3) | 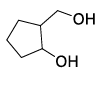 |
| (S) | 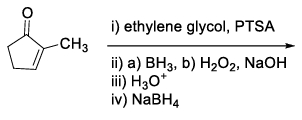 | (4) | 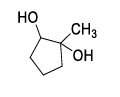 |
| (5) |  | ||
First write down the complete data of the question.
List-I (reaction sequences):
P. $$C_6H_5OCH_3 \;\xrightarrow[\text{excess}]{\,HI\,}$$ (anisole is heated with excess hydroiodic acid)
Q. $$C_6H_5OH \;\xrightarrow[]{\,conc.\;HNO_3/\,conc.\;H_2SO_4\,}$$ (phenol treated with conc. nitrating mixture)
R. $$C_6H_5OH \;\xrightarrow[]{\,CH_3COCl/pyridine\,}$$ (phenol acetylated with acetyl chloride in pyridine)
S. $$C_6H_5OH \;\xrightarrow[]{\,NaOH,\;140^{\circ}C,\;7\,atm\,}\;NaO\!C_6H_4O^-$$ $$\xrightarrow[]{\,CO_2\,}\xrightarrow[]{\,H^+\,}$$ (Kolbe-Schmitt reaction followed by acidification)
List-II (probable products):
1. $$o\text{-}HO\!C_6H_4COOH$$ (salicylic acid)
2. $$p\text{-}HO\!C_6H_4NO_2$$ (p-nitrophenol)
3. $$C_6H_5OH + CH_3I$$
4. $$C_6H_5OCOCH_3$$ (phenyl acetate)
5. $$2,4,6\text{-}trinitrophenol$$ (picric acid)
Now analyse every reaction one by one.
Case P:Anisole $$\left(C_6H_5OCH_3\right)$$ when heated with excess $$HI$$ undergoes cleavage of the C-O bond attached to the alkyl group, forming phenol and methyl iodide: $$C_6H_5OCH_3 + HI \;\longrightarrow\; C_6H_5OH + CH_3I$$ This matches product 3.
Case Q:Phenol reacts with concentrated $$HNO_3/H_2SO_4$$ at low temperature to give picric acid (2,4,6-trinitrophenol) because -OH is a strong activator and three nitro groups finally enter the ring: $$C_6H_5OH \xrightarrow[]{conc.\;HNO_3/H_2SO_4} 2,4,6\text{-}trinitrophenol$$ Hence Q corresponds to product 5.
Case R:Acetylation of phenol with acetyl chloride in pyridine gives phenyl acetate: $$C_6H_5OH + CH_3COCl \;\xrightarrow[]{pyridine}\; C_6H_5OCOCH_3 + HCl$$ Therefore R matches product 4.
Case S:The Kolbe-Schmitt reaction converts phenoxide ion to o-hydroxybenzoate ion which after acidification yields salicylic acid: $$C_6H_5OH \xrightarrow[]{NaOH} C_6H_5O^-Na^+ \xrightarrow[7\,atm]{CO_2,\;140^{\circ}C} o\text{-}HO\!C_6H_4COONa \xrightarrow[]{H^+} o\text{-}HO\!C_6H_4COOH$$ So S corresponds to product 1.
Collecting all matches:
P → 3, Q → 5, R → 4, S → 1
The option that contains this combination is Option A.
Final answer: Option A which is: P-3, Q-5, R-4, S-1.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
