Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In a conductometric titration, small volume of titrant of higher concentration is added stepwise to a larger volume of titrate of much lower concentration, and the conductance is measured after each addition.
The limiting ionic conductivity ($$\Lambda_0$$) values (in mS $$m^2$$ $$mol^{-1}$$) for different ions in aqueous solutions are given below:
| Ions | $$Ag^+$$ | $$K^+$$ | $$Na^+$$ | $$H^+$$ | $$NO_3^-$$ | $$Cl^-$$ | $$SO_4^{2-}$$ | $$OH^-$$ | $$CH_3COO^-$$ |
|---|---|---|---|---|---|---|---|---|---|
| $$\Lambda_0$$ | 6.2 | 7.4 | 5.0 | 35.0 | 7.2 | 7.6 | 16.0 | 19.9 | 4.1 |
For different combinations of titrates and titrants given in List-I, the graphs of 'conductance' versus 'volume of titrant' are given in List-II.
Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | Titrate: KCl Titrant: $$AgNO_3$$ | (1) | 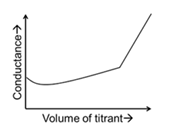 |
| (Q) | Titrate: $$AgNO_3$$ Titrant: KCl | (2) | 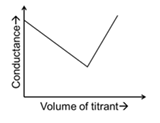 |
| (R) | Titrate: NaOH Titrant: HCl | (3) | 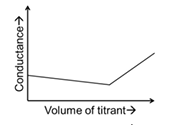 |
| (S) | Titrate: NaOH Titrant: $$CH_3COOH$$ | (4) | 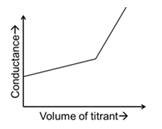 |
| (5) | 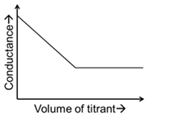 | ||
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
