Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In the following reaction sequence, the major product P is formed.
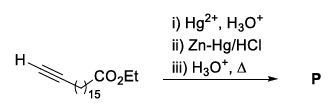
Glycerol reacts completely with excess P in the presence of an acid catalyst to form Q. Reaction of Q with excess NaOH followed by the treatment with $$CaCl_2$$ yields Ca-soap R, quantitatively. Starting with one mole of Q, the amount of R produced in gram is ______.
[Given, atomic weight: H = 1, C = 12, N = 14, O = 16, Na = 23, Cl = 35, Ca = 40]
Correct Answer: 909
Let the major product $$P$$ obtained in the earlier sequence be stearic acid, $$C_{17}H_{35}COOH$$.
With an acid catalyst, glycerol esterifies completely with excess stearic acid to give the tri-ester (a triglyceride) $$Q$$, called tristearin.
Step-1 : Formation of $$Q$$
Glycerol possesses three -OH groups, so three molecules of stearic acid react with one molecule of glycerol and three molecules of water are eliminated:
$$C_3H_8O_3 + 3\,C_{17}H_{35}COOH \;\xrightarrow{\text{acid}}\; C_3H_5\big(OOC\,C_{17}H_{35}\big)_3 + 3\,H_2O$$
Molar mass of glycerol $$= 3\times12 + 8\times1 + 3\times16 = 92$$ g mol$$^{-1}$$
Molar mass of stearic acid $$= 18\times12 + 36\times1 + 2\times16 = 284$$ g mol$$^{-1}$$
Molar mass of $$Q$$ (tristearin):
$$M_Q = 92 + 3(284) - 3(18) = 92 + 852 - 54 = 890\;\text{g mol}^{-1}$$
Step-2 : Alkaline hydrolysis (saponification) of $$Q$$ with excess $$NaOH$$
$$C_3H_5\big(OOC\,C_{17}H_{35}\big)_3 + 3\,NaOH \;\rightarrow\; 3\,C_{17}H_{35}COO^-Na^+ + C_3H_8O_3$$
Thus 1 mol of $$Q$$ gives 3 mol of sodium stearate.
Molar mass of sodium stearate:
Carboxylate anion $$C_{17}H_{35}COO^-$$ has mass $$284 - 1 = 283$$ g.
Adding Na (23 g) gives $$M_{Na\text{-stearate}} = 283 + 23 = 306\;\text{g mol}^{-1}$$
Step-3 : Conversion of sodium stearate to calcium stearate (Ca-soap $$R$$) using excess $$CaCl_2$$
$$2\,C_{17}H_{35}COO^-Na^+ + CaCl_2 \;\rightarrow\; (C_{17}H_{35}COO^-)_2Ca^{2+} + 2\,NaCl$$
Stoichiometry: 2 mol Na-stearate → 1 mol Ca-stearate.
From Step-2 we have 3 mol Na-stearate, therefore
Number of moles of $$R$$ obtained
$$= \frac{3}{2} = 1.5\;\text{mol}$$
Molar mass of calcium stearate $$R$$:
$$M_R = 40 + 2(283) = 40 + 566 = 606\;\text{g mol}^{-1}$$
Mass of $$R$$ from 1 mol of $$Q$$:
$$m_R = 1.5 \times 606 = 909\;\text{g}$$
Therefore, starting with one mole of $$Q$$, the amount of calcium soap $$R$$ produced is 909 g.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
