Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In figure, a straight line is given for Freundrich Adsorption ($$y = 3x + 2.505$$). The value of $$\frac{1}{n}$$ and log K are respectively.
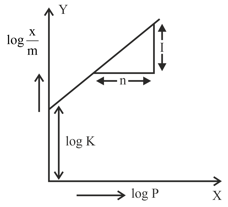
We are given the Freundlich adsorption isotherm plotted as a straight line: $$y = 3x + 2.505$$.
The Freundlich equation is: $$\frac{x}{m} = K \cdot p^{1/n}$$
Taking logarithm:
$$\log\frac{x}{m} = \log K + \frac{1}{n}\log p$$
Comparing with $$y = 3x + 2.505$$:
Here $$y = \log(x/m)$$ and $$x$$-axis = $$\log p$$.
Slope = $$\frac{1}{n} = 3$$
Intercept = $$\log K = 2.505$$
$$\frac{1}{n} = 3$$ and $$\log K = 2.505$$.
The correct answer is Option C: $$3$$ and $$2.505$$.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
