Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
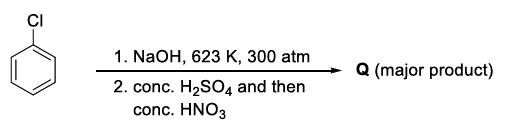
The weight percentage of hydrogen in Q, formed in the following reaction sequence, is ______.
[Given : Atomic mass of H = 1, C = 12, N = 14, O = 16, S = 32, Cl = 35]
Correct Answer: 1.31
The reaction sequence (conc. $$H_2SO_4$$ followed by conc. $$HNO_3$$) converts phenol first into its sulphonic-acid derivative and finally into 2,4,6-trinitrophenol (picric acid). Hence the compound $$Q$$ is 2,4,6-trinitrophenol having the molecular formula $$C_6H_3N_3O_7$$.
Step 1 : Find the molecular mass of $$Q$$.
$$\begin{aligned}
M &= 6\times12 \;(\text{for }C) + 3\times1 \;(\text{for }H) + 3\times14 \;(\text{for }N) + 7\times16 \;(\text{for }O) \\
&= 72 + 3 + 42 + 112 \\
&= 229 \text{ g mol}^{-1}
\end{aligned}$$
Step 2 : Calculate the mass percentage of hydrogen.
Number of hydrogen atoms = 3, so mass of hydrogen in one mole = $$3\times1 = 3$$ g.
Weight % of H $$= \dfrac{\text{mass of H}}{\text{molar mass}}\times100$$
$$\frac{3}{229}\times100 = 1.31\%$$
Thus, the weight percentage of hydrogen in $$Q$$ is 1.31 %.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
