Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Arrange in the correct order of stability (decreasing order) for the following molecules: (I), (II), (III), (IV) - cyclohexane conformations
I.
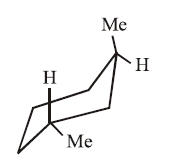
II.
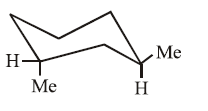
III.
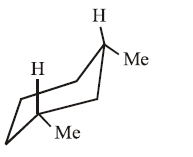
IV.
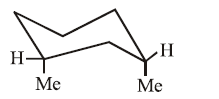
Molecule (III): Both methyl groups are in equatorial positions. This diequatorial arrangement minimizes steric repulsion, making it the most stable conformation in the set.
Molecules (I) and (II): In both structures, one methyl group is equatorial and the other is axial. Because each contains exactly one axial group causing 1,3-diaxial interaction, their energy levels, and thus their stabilities are approximately equal.
Molecule (IV): Both methyl groups are in axial positions. This diaxial arrangement creates significant steric strain (1,3-diaxial interactions) from both groups simultaneously, making it the least stable conformation.
Stability: $$(III) > (I) \approx (II) > (IV)$$
Create a FREE account and get:
Educational materials for JEE preparation
