Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Which of the following represents the Freundlich adsorption isotherms?
(A)
(B)
(C)
(D)
Choose the correct answer from the options given below:
The Freundlich adsorption isotherm is: $$\frac{x}{m} = kP^{1/n}$$
This can be represented graphically in several ways:
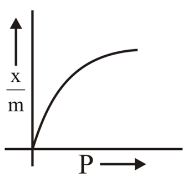
A. $$x/m$$ vs $$P$$: This gives a curve (not linear) which is characteristic of Freundlich adsorption. ✓
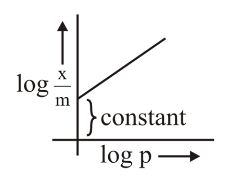
B. Taking log on both sides: $$\log\frac{x}{m} = \log k + \frac{1}{n}\log P$$. Plot of $$\log(x/m)$$ vs $$\log P$$ is a straight line with slope $$1/n$$ and intercept $$\log k$$. ✓
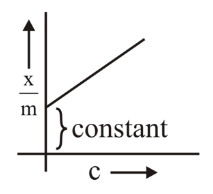
C. $$x/m$$ vs $$c$$ (concentration): This doesn't directly represent the Freundlich isotherm equation (which uses pressure for gases). ✗
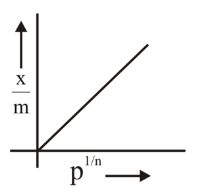
D. $$x/m$$ vs $$P^{1/n}$$: From the equation, $$x/m = k \cdot P^{1/n}$$, this is a straight line passing through the origin with slope $$k$$. ✓
The correct representations are A, B, D only.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.