Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In an acid-base titration, 0.1 M HCl solution was added to the NaOH solution of unknown strength. Which of the following correctly shows the change of pH of the titration mixture in this experiment?
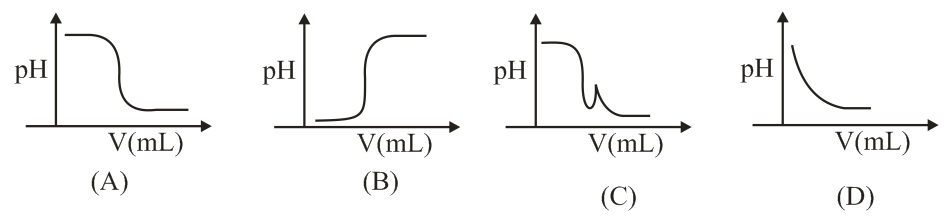
At the start of the experiment we have only the sodium hydroxide solution in the conical flask. Sodium hydroxide is a strong base, so it dissociates completely according to
$$\mathrm{NaOH \;\longrightarrow\; Na^+ + OH^-}$$
Because this dissociation is complete, the initial concentration of hydroxide ion equals the initial molarity of the NaOH solution. Although the exact molarity of this NaOH is unknown, it is certainly greater than zero, so the initial value of $$[\text{OH}^-]$$ is appreciable and the pH is therefore high. Using the relation
$$\text{pOH} \;=\; -\log_{10} [\text{OH}^-]$$
and the identity
$$\text{pH} + \text{pOH} = 14$$
we see that when $$[\text{OH}^-]$$ is large, pOH becomes small and pH becomes large, giving an initial pH close to 13-14. Hence the titration curve must begin in the high-pH region.
We now begin to add 0.1 M hydrochloric acid from the burette. Hydrochloric acid is a strong acid and dissociates completely:
$$\mathrm{HCl \;\longrightarrow\; H^+ + Cl^-}$$
Each added $$\mathrm{H^+}$$ ion reacts instantaneously and quantitatively with an $$\mathrm{OH^-}$$ ion present in the flask according to
$$\mathrm{H^+ + OH^- \;\longrightarrow\; H_2O}$$
Thus, as we go on adding small volumes $$\Delta V$$ of the 0.1 M acid, the number of moles of hydroxide ion in the flask decreases step by step while the total volume of the mixture increases. The working expression after each small addition is
$$n_{\text{OH}^-}^{\text{remaining}} \;=\; n_{\text{OH}^-}^{\text{initial}} \;-\; n_{\text{H}^+}^{\text{added}}$$
and, because volume is changing, the hydroxide concentration becomes
$$[\text{OH}^-] \;=\; \dfrac{n_{\text{OH}^-}^{\text{remaining}}}{V_{\text{initial}} + V_{\text{added}}}$$
Whenever $$n_{\text{OH}^-}^{\text{remaining}}\gt 0$$ we still have a basic solution, so pH stays > 7, but because both the numerator (moles of OH⁻) is falling and the denominator (total volume) is rising, the concentration $$[\text{OH}^-]$$ - and hence the pH - falls gradually. Therefore the left-hand part of the titration curve must show a gentle downward slope from the initial high pH.
A particular volume of acid, which we call the equivalence volume $$V_\text{eq}$$, will exactly neutralise the original moles of NaOH, i.e.
$$n_{\text{H}^+}^{\text{added at }V_\text{eq}} \;=\; n_{\text{OH}^-}^{\text{initial}}$$
Right at this equivalence point we have neither free hydroxide ion nor free hydrogen ion in solution; instead we have a solution that contains only the neutral salt $$\mathrm{NaCl}$$. Because both the acid and the base are strong, the pH at the equivalence point is governed solely by the auto-ionisation of water:
$$\mathrm{H_2O \;\rightleftharpoons\; H^+ + OH^-} \quad\text{with}\quad K_\mathrm{w} = 1.0 \times 10^{-14}$$
The solution is therefore neutral, giving
$$[\text{H}^+] = [\text{OH}^-] = 1.0 \times 10^{-7}\,\text{M}, \quad\text{so}\quad \text{pH} = 7$$
Because a very small additional amount of acid beyond $$V_\text{eq}$$ produces an excess of hydrogen ion in a large, but not enormous, total volume, the hydrogen-ion concentration rises sharply and the pH drops abruptly. Mathematically, just after equivalence we have
$$[\text{H}^+] = \dfrac{n_{\text{H}^+}^{\text{excess}}}{V_{\text{total}}}$$
where $$n_{\text{H}^+}^{\text{excess}}$$ is tiny, but non-zero, and $$V_{\text{total}} \approx V_{\text{initial}} + V_\text{eq}$$. Even a few microlitres of excess strong acid in a near-neutral solution push the pH down by several units. Hence the titration curve shows a very steep vertical fall centred on pH 7.
After a substantial excess of acid has been added, the hydroxide ion concentration becomes negligible compared with the directly calculated hydrogen-ion concentration, so the pH approaches that of the added 0.1 M HCl, which is
$$\text{pH} = -\log_{10}(0.1) = 1$$
Consequently, the right-hand tail of the curve flattens out in the low-pH region near pH 1.
Putting the entire story together, the correct titration curve must
1. start at a very high pH (>13),
2. decrease slowly as acid is added,
3. show a sudden, nearly vertical drop at the equivalence volume, passing through pH 7, and
4. level off near pH 1 when a large excess of acid is present.
Among the options given, only diagram A exhibits exactly this pattern: a gentle fall from high pH, a sharp vertical step at pH 7, and a low-pH plateau. Therefore option A matches the expected behaviour for the titration of an unknown-strength NaOH with 0.1 M HCl.
Hence, the correct answer is Option A.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.