Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Henry's constant (in kbar) for four gases $$\alpha$$, $$\beta$$, $$\gamma$$ and $$\delta$$ in water at 298 K is given below:
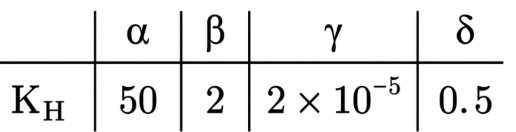
(density of water = 10$$^3$$ kg m$$^{-3}$$ at 298 K) This table implies that:
For a gas dissolved in a liquid Henry’s law is stated as
$$p = K_H\,x$$
where $$p$$ is the partial pressure of the gas (in the same units as $$K_H$$) and $$x$$ is the mole-fraction of the dissolved gas in the solution. From the equation we immediately notice that, for any fixed pressure, a smaller value of $$K_H$$ must give a larger value of $$x$$; therefore the smaller the Henry’s constant, the greater is the solubility of the gas.
Checking option A
The constants are $$K_H(\alpha)=50\ \text{kbar},\;K_H(\beta)=2\ \text{kbar},\;K_H(\gamma)=2\times10^{-5}\ \text{kbar},\;K_H(\delta)=0.5\ \text{kbar}.$$
Because 50 kbar is the largest value, $$\alpha$$ actually has the least solubility, not the greatest. So option A is wrong.
Checking option B
No numerical value of $$K_H$$ at 308 K has been supplied, and the question does not state whether dissolution of $$\gamma$$ is exothermic (for which $$K_H$$ normally rises with temperature) or endothermic (for which it would fall). Thus we cannot deduce the solubility change only from the given data. Hence option B cannot be affirmed and is taken as incorrect.
Checking option C
A 55.5 molal solution means 55.5 mol of the gas dissolved per 1 kg of water. Because the molar mass of water is $$18\ \text{g mol}^{-1}$$, 1 kg of water contains
$$n_{\text{water}}=\frac{1000\ \text{g}}{18\ \text{g mol}^{-1}}=55.5\ \text{mol}.$$
The dissolved gas also has $$n_{\text{gas}}=55.5\ \text{mol}.$$ Hence the total number of moles in the solution is
$$n_{\text{total}}=n_{\text{gas}}+n_{\text{water}}=55.5+55.5=111\ \text{mol}.$$
The mole-fraction of the gas is therefore
$$x_{\delta}=\frac{n_{\text{gas}}}{n_{\text{total}}}=\frac{55.5}{111}=0.5.$$
Using Henry’s law with $$K_H(\delta)=0.5\ \text{kbar}$$, we have
$$p_{\delta}=K_H\,x_{\delta}=0.5\ \text{kbar}\times0.5=0.25\ \text{kbar}.$$
Because $$1\ \text{kbar}=1000\ \text{bar},$$ we convert
$$p_{\delta}=0.25\ \text{kbar}=0.25\times1000\ \text{bar}=250\ \text{bar}.$$
This exactly matches the value stated in option C, so option C is correct.
Checking option D
With the same 55.5 molal concentration we have the same mole-fraction $$x_{\gamma}=0.5.$$
For $$\gamma$$, $$K_H(\gamma)=2\times10^{-5}\ \text{kbar}.$$
Applying Henry’s law gives
$$p_{\gamma}=K_H\,x_{\gamma}=(2\times10^{-5}\ \text{kbar})\times0.5=1\times10^{-5}\ \text{kbar}.$$
Converting to bar,
$$p_{\gamma}=1\times10^{-5}\ \text{kbar}\times1000\ \text{bar kbar}^{-1}=0.01\ \text{bar}.$$
This is far lower than 1 bar, so option D is false.
Among the four statements, only option C is consistent with Henry’s law and the numerical data.
Hence, the correct answer is Option C.
Create a FREE account and get:
Educational materials for JEE preparation
