Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
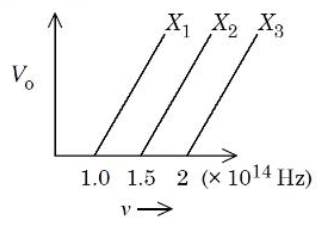
The graph shows variation of stopping potential $$V_0$$ with the frequency $$\nu$$ of the incident radiation for three photosensitive metals $$X_1$$, $$X_2$$ and $$X_3$$. Which metal will give out electrons with greater kinetic energy, for the same wavelength of incident radiation?
The photoelectric equation relates the maximum kinetic energy of the emitted electrons to the stopping potential:
$$\text{K.E.}_{\max} = eV_0 = h\nu - \phi$$
where
• $$h$$ is Planck’s constant,
• $$\nu$$ is the frequency of the incident radiation,
• $$\phi$$ is the work function of the metal,
• $$e$$ is the electronic charge.
Re-writing it in the form of a straight line:
$$V_0 = \frac{h}{e}\,\nu - \frac{\phi}{e} \qquad -(1)$$
Equation $$(1)$$ shows that a plot of $$V_0$$ (vertical axis) versus $$\nu$$ (horizontal axis) is a straight line with
• slope $$\frac{h}{e}$$ (same for every metal),
• intercept on the $$V_0$$-axis equal to $$-\frac{\phi}{e}$$, and
• intercept on the $$\nu$$-axis (threshold frequency) $$\nu_0 = \frac{\phi}{h}$$.
For a fixed incident wavelength (that is, a fixed frequency $$\nu$$ common to all three metals), the maximum kinetic energy is directly proportional to the stopping potential $$V_0$$. Hence, the metal that exhibits the largest $$V_0$$ at that chosen $$\nu$$ will emit the electrons with the greatest kinetic energy.
Because the slope is identical for all three lines, the line lying highest on the graph at any given $$\nu$$ is the one whose $$\nu$$-axis intercept $$\nu_0$$ is the smallest (lowest threshold frequency). A smaller $$\nu_0$$ means a smaller work function $$\phi$$, so the electrons require less energy to be liberated and therefore retain more kinetic energy.
Among the three plotted lines, metal $$X_1$$ has the lowest threshold frequency (its line meets the $$\nu$$-axis farthest to the left). Consequently, for the same incident frequency (or wavelength) it gives the largest stopping potential $$V_0$$ and, therefore, the greatest maximum kinetic energy of the emitted electrons.
Hence, the required metal is:
Option A which is: $$X_1$$
Create a FREE account and get:
Educational materials for JEE preparation
