Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the $$P$$-$$V$$ diagrams below. In cycle I, processes a, b, c and d are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes a', b', c' and d' are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is $$W_I$$ and that during cycle II is $$W_{II}$$. The ratio $$W_I/W_{II}$$ is ____.
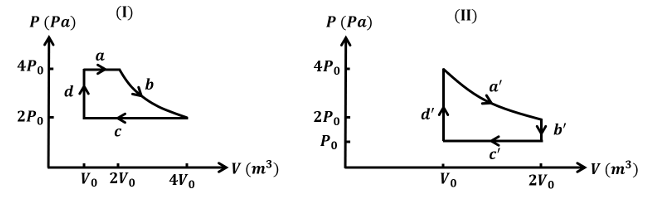
Correct Answer: 2
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
