Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
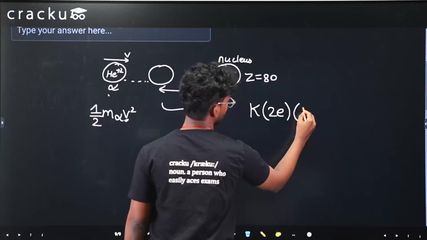
In an alpha particle scattering experiment distance of closest approach for the $$\alpha$$ particle is $$4.5 \times 10^{-14} \text{ m}$$. If target nucleus has atomic number 80, then maximum velocity of $$\alpha$$-particle is ______ $$\times 10^5 \text{ m/s}$$ approximately. $$\left(\frac{1}{4\pi\epsilon_0} = 9 \times 10^9 \text{ SI unit, mass of } \alpha \text{ particle} = 6.72 \times 10^{-27} \text{ kg}\right)$$
Correct Answer: 156
We need to find the maximum velocity of an alpha particle in a scattering experiment.
At the distance of closest approach, all kinetic energy is converted to electrostatic potential energy:
$$\frac{1}{2}mv^2 = \frac{1}{4\pi\epsilon_0}\frac{(2e)(Ze)}{d}$$
$$Z = 80$$, $$d = 4.5 \times 10^{-14}$$ m, $$m = 6.72 \times 10^{-27}$$ kg, $$e = 1.6 \times 10^{-19}$$ C, $$\frac{1}{4\pi\epsilon_0} = 9 \times 10^9$$.
$$\frac{1}{2}mv^2 = \frac{9 \times 10^9 \times 2 \times 80 \times (1.6 \times 10^{-19})^2}{4.5 \times 10^{-14}}$$
$$9 \times 10^9 \times 160 \times 2.56 \times 10^{-38} = 9 \times 160 \times 2.56 \times 10^{-29}$$
$$= 9 \times 409.6 \times 10^{-29} = 3686.4 \times 10^{-29} = 3.6864 \times 10^{-26}$$
$$\frac{1}{2}mv^2 = \frac{3.6864 \times 10^{-26}}{4.5 \times 10^{-14}} = 8.192 \times 10^{-13}$$ J
$$v^2 = \frac{2 \times 8.192 \times 10^{-13}}{6.72 \times 10^{-27}} = \frac{16.384 \times 10^{-13}}{6.72 \times 10^{-27}} = 2.438 \times 10^{14}$$
$$v = \sqrt{2.438 \times 10^{14}} \approx 1.56 \times 10^7 \text{ m/s} = 156 \times 10^5$$ m/s
The maximum velocity is approximately $$156 \times 10^5$$ m/s.
Therefore, the answer is 156.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
