Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The left and right compartments of a thermally isolated container of length $$L$$ are separated by a thermally conducting, movable piston of area $$A$$. The left and right compartments are filled with $$\frac{3}{2}$$ and 1 moles of an ideal gas, respectively. In the left compartment the piston is attached to a spring with spring constant $$k$$ and natural length $$\frac{2L}{5}$$. In thermodynamic equilibrium, the piston is at a distance $$\frac{L}{2}$$ from the left and right edges of the container as shown in the figure. Under the above conditions, if the pressure in the right compartment is $$P = \frac{kL}{A} \alpha$$, then the value of $$\alpha$$ is ______.
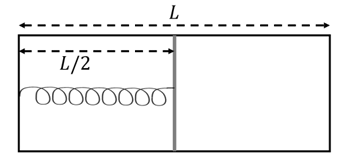
Correct Answer: 0.2
Let the number of moles, the pressure and the volume in the left compartment be $$n_1,\;P_L,\;V_L$$ and in the right compartment be $$n_2,\;P_R,\;V_R$$.
Given
$$n_1 = \frac32,\qquad n_2 = 1$$
length of each compartment at equilibrium: $$\frac{L}{2}$$
cross-sectional area of the piston: $$A$$
Therefore
$$V_L = A \left(\frac{L}{2}\right)=V_R = A\left(\frac{L}{2}\right)=V$$ where $$V = \frac{AL}{2}\,.$$
The piston is thermally conducting, hence the two gases finally attain the same temperature $$T$$. For an ideal gas, $$PV=nRT$$; therefore
$$P_LV = n_1 RT,\qquad P_RV = n_2 RT$$
Dividing the first relation by the second,
$$\frac{P_L}{P_R} = \frac{n_1}{n_2}= \frac{3/2}{1}= \frac32 \quad\Longrightarrow\quad P_L = \frac32\,P_R\;.$$ -(1)
Next, write the force-balance for the movable piston. Choose the rightward direction as positive.
• Force on piston by left gas (to the right): $$P_LA$$
• Force on piston by right gas (to the left): $$P_RA$$
• Force by the spring (to the left): $$F_s$$
At mechanical equilibrium, net force is zero:
$$P_LA - P_RA - F_s = 0 \quad\Longrightarrow\quad (P_L-P_R)A = F_s\;.$$ -(2)
The spring is fixed to the left wall; its natural length is $$\dfrac{2L}{5}$$ and its actual length equals the left-hand length $$\dfrac{L}{2}$$. Hence the extension is
$$\Delta x = \frac{L}{2} - \frac{2L}{5} = \frac{L}{10}\;.$$
Thus the spring force is
$$F_s = k\,\Delta x = k\left(\frac{L}{10}\right)=\frac{kL}{10}\;.$$ -(3)
Insert $$(1)$$ and $$(3)$$ into $$(2)$$:
$$(P_L-P_R)A = \frac{kL}{10}$$ $$\left(\frac32\,P_R - P_R\right)A = \frac{kL}{10}$$ $$\left(\frac12\,P_R\right)A = \frac{kL}{10}$$ $$P_R = \frac{kL}{10A}\times 2 = \frac{kL}{5A}\;.$$
Therefore the pressure in the right compartment is
$$P = P_R = \frac{kL}{A}\left(\frac15\right)\,.$$
Comparing with the required form $$P = \dfrac{kL}{A}\,\alpha$$ gives
$$\alpha = \frac15 = 0.2\;.$$
Hence, the desired value is 0.2.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
