Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A cylindrical tube $$AB$$ of length $$l$$, closed at both ends contains an ideal gas of 1 mol having molecular weight $$M$$. The tube is rotated in a horizontal plane with constant angular velocity $$\omega$$ about an axis pe1pendicular to $$AB$$ and passing through the edge at end $$A$$ , as shown in the figure. If $$P_{A}$$ and $$P_{B}$$ are the pressures at $$A$$ and $$B$$ respectively, then
(Consider the temperature is same at all points in the tube)
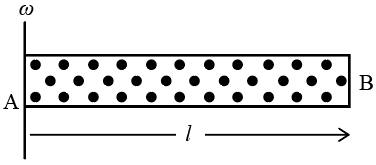
Consider a small gas element at distance r from the axis at end A.
Because the tube rotates with angular speed ω\omegaω, that gas element needs centripetal force.
Required centripetal force on volume element dV=A dr is
$$dmω^2r$$
where
$$dm=ρAdr$$
So force balance due to pressure difference gives
$$dP⋅A=ρAdrω^2r$$
Cancel A:
$$dP=ρω^2rdr$$
For an ideal gas,
$$\rho=\frac{MP}{RT}$$
(M is molar mass, one mole gas)
Substitute:
$$dP=\frac{MP}{RT}ω^2rdr$$
Rearrange:
$$\frac{dP}{P}=\frac{M}{RT}ω^2rdr$$
Integrate from end A to end B:
At
$$r=0,\quad P=P_A$$
At
$$r=l,\quad P=P_B$$
So
$$\int_{P_A}^{P_B}\frac{dP}{P}=\frac{M\omega^2}{RT}\int\ rdr$$
$$\ln\frac{P_B}{P_A}=\frac{M\omega^2}{RT}\cdot\frac{l^2}{2}l$$
Which is same as
$$P_{B}=P_{A} exp(M \omega^{2}l^{2}/2RT)$$
Create a FREE account and get:
Educational materials for JEE preparation
