Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
One mole of an ideal gas at 27 $$^\circ$$C is taken from A to B as shown in the given PV indicator diagram. The work done by the system will be ___ $$\times 10^{-1}$$ J. [Given, $$R = 8.3$$ J mole$$^{-1}$$ K, ln 2 = 0.6931] (Round off to the nearest integer)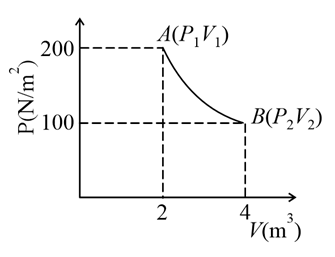
Correct Answer: 17258
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.