Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
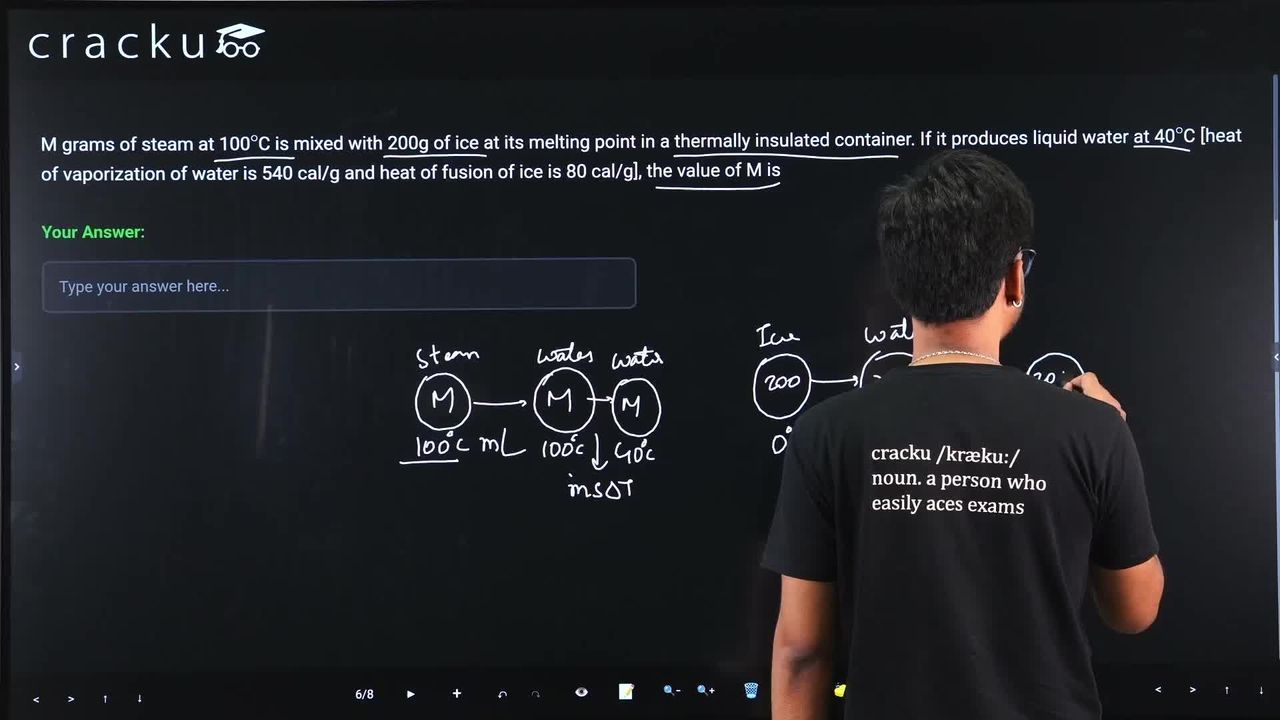
M grams of steam at 100$$^\circ$$C is mixed with 200g of ice at its melting point in a thermally insulated container. If it produces liquid water at 40$$^\circ$$C [heat of vaporization of water is 540 cal/g and heat of fusion of ice is 80 cal/g], the value of M is
Correct Answer: 40
We begin by noting that the container is perfectly insulated, so the total heat released by the hot substance (steam) must equal the total heat absorbed by the cold substance (ice). In symbols, we write $$Q_{\text{released}} = Q_{\text{absorbed}}.$$
Let $$M$$ grams be the mass of steam at $$100^\circ\text{C}$$ that is mixed with $$200\ \text{g}$$ of ice at $$0^\circ\text{C}$$. Eventually the whole system reaches $$40^\circ\text{C}$$ as liquid water.
We treat the heat changes in two separate stages for each substance.
1. Heat released by the steam
(a) First, the steam condenses to water at the same temperature $$100^\circ\text{C}$$. The formula for heat involved in a phase change is $$Q = mL,$$ where $$m$$ is the mass and $$L$$ is the latent heat. For condensation we use the latent heat of vaporization of water, $$L_v = 540\ \text{cal g}^{-1}.$$ Hence the heat released during condensation is $$Q_1 = M \times 540.$$
(b) Next, the resulting water cools from $$100^\circ\text{C}$$ down to the final temperature $$40^\circ\text{C}$$. The formula for heat exchange during a temperature change is $$Q = mc\Delta T,$$ where $$c$$ is the specific heat capacity. For liquid water, $$c = 1\ \text{cal g}^{-1\ ^\circ\!C}$$. The temperature drop is $$\Delta T = 100 - 40 = 60^\circ\text{C}.$$ Thus the heat released during cooling is $$Q_2 = M \times 1 \times 60 = 60M.$$
Total heat released by the steam is therefore $$Q_{\text{released}} = Q_1 + Q_2 = 540M + 60M = 600M.$$
2. Heat absorbed by the ice
(a) First, the ice melts at $$0^\circ\text{C}$$ to become water at the same temperature. Using $$Q = mL$$ with the latent heat of fusion $$L_f = 80\ \text{cal g}^{-1}$$, we have $$Q_3 = 200 \times 80 = 16\,000.$$
(b) Next, the melt-water warms from $$0^\circ\text{C}$$ to $$40^\circ\text{C}$$. Using $$Q = mc\Delta T$$ with $$\Delta T = 40^\circ\text{C}$$, we get $$Q_4 = 200 \times 1 \times 40 = 8\,000.$$
Total heat absorbed by the ice and cold water is $$Q_{\text{absorbed}} = Q_3 + Q_4 = 16\,000 + 8\,000 = 24\,000.$$
3. Applying energy conservation
Since no heat is lost to the surroundings, we equate the two totals: $$Q_{\text{released}} = Q_{\text{absorbed}},$$ $$600M = 24\,000.$$
Solving for $$M$$ gives $$M = \frac{24\,000}{600} = 40.$$
Thus, $$40\ \text{g}$$ of steam are required.
So, the answer is $$40$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
