Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
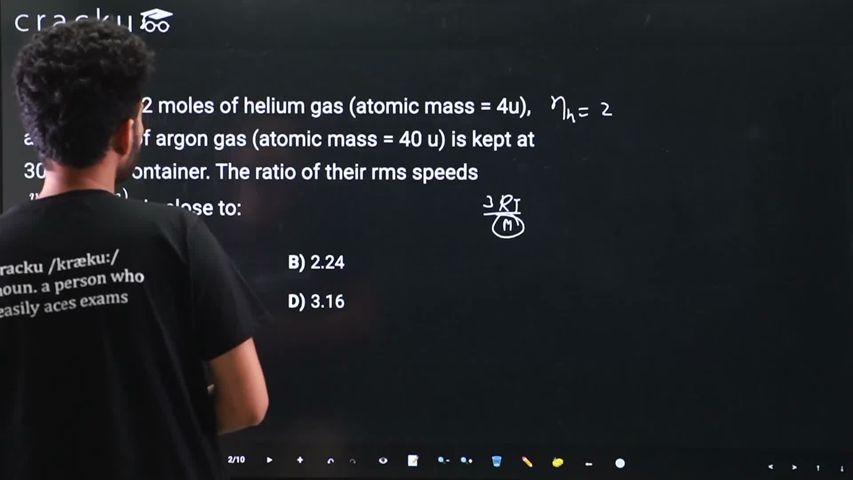
A mixture of 2 moles of helium gas (atomic mass = 4u), and 1 mole of argon gas (atomic mass = 40 u) is kept at 300 K in a container. The ratio of their rms speeds $$\frac{v_{rms}(helium)}{v_{rms}(argon)}$$, is close to:
For any ideal gas, the root-mean-square (rms) speed of its molecules is given by the formula
$$v_{rms}=\sqrt{\frac{3RT}{M}},$$
where $$R$$ is the universal gas constant, $$T$$ is the absolute temperature in kelvin, and $$M$$ is the molar mass of the gas in kilograms per mole. This expression follows directly from equating the kinetic-theory expression for pressure with the ideal-gas law.
We are asked to compare the rms speeds of helium and argon at the same temperature. Because $$R$$ and $$T$$ are identical for both gases, the ratio of their rms speeds will depend only on the inverse square root of their molar masses. Mathematically, we can write
$$\frac{v_{rms}(\text{helium})}{v_{rms}(\text{argon})} =\sqrt{\frac{3RT/M_{\text{He}}}{3RT/M_{\text{Ar}}}} =\sqrt{\frac{M_{\text{Ar}}}{M_{\text{He}}}}.$$
Now we substitute the numerical molar masses. The atomic (and hence molar) mass of helium is given as $$4\ \text{u}=4\ \text{g mol}^{-1}$$, while that of argon is $$40\ \text{u}=40\ \text{g mol}^{-1}$$. Converting grams per mole to kilograms per mole is not necessary for a ratio because the factor of $$10^{-3}$$ cancels out, but we can still show it explicitly:
$$M_{\text{He}} = 4\ \text{g mol}^{-1}=4\times10^{-3}\ \text{kg mol}^{-1},$$
$$M_{\text{Ar}} = 40\ \text{g mol}^{-1}=40\times10^{-3}\ \text{kg mol}^{-1}.$$
Substituting these values into the ratio expression, we obtain
$$\frac{v_{rms}(\text{He})}{v_{rms}(\text{Ar})} =\sqrt{\frac{40\times10^{-3}}{4\times10^{-3}}} =\sqrt{\frac{40}{4}} =\sqrt{10} \approx 3.162.$$
This numerical value is closest to $$3.16$$ in the given options.
Hence, the correct answer is Option D.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
