Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Three Carnot engines operate in series between a heat source at a temperature $$T_1$$ and a heat sink at temperature $$T_4$$ (see figure). There are two other reservoirs at temperature $$T_2$$ and $$T_3$$, as shown, with $$T_1 > T_2 > T_3 > T_4$$. The three engines are equally efficient if:
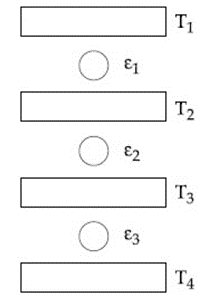
We are given three Carnot engines working one after the other (in series). The temperatures of the four reservoirs, arranged from hottest to coldest, are $$T_1, T_2, T_3,$$ and $$T_4$$ with $$T_1 > T_2 > T_3 > T_4.$$
For any Carnot engine operating between a hot reservoir at temperature $$T_h$$ and a cold reservoir at $$T_c,$$ the thermal efficiency is, by definition,
$$\eta = 1-\dfrac{T_c}{T_h}.$$
Because the engines are in series, the first engine works between $$T_1$$ (hot) and $$T_2$$ (cold), the second between $$T_2$$ and $$T_3,$$ and the third between $$T_3$$ and $$T_4.$$ Their efficiencies are therefore
$$\eta_1 = 1-\dfrac{T_2}{T_1}, \qquad \eta_2 = 1-\dfrac{T_3}{T_2}, \qquad \eta_3 = 1-\dfrac{T_4}{T_3}.$$
The statement “the three engines are equally efficient’’ means
$$\eta_1 = \eta_2 = \eta_3.$$
We first equate $$\eta_1$$ and $$\eta_2.$$ Starting with the equality
$$1-\dfrac{T_2}{T_1} = 1-\dfrac{T_3}{T_2},$$
we cancel the 1’s on both sides, giving
$$-\dfrac{T_2}{T_1} = -\dfrac{T_3}{T_2}.$$
Multiplying through by $$-1$$ to remove the negative sign, we have
$$\dfrac{T_2}{T_1} = \dfrac{T_3}{T_2}.$$
Cross-multiplication now yields
$$T_2^2 = T_1\,T_3,$$
and solving for $$T_3$$ gives
$$T_3 = \dfrac{T_2^2}{T_1}. \quad -(1)$$
Next we equate $$\eta_2$$ and $$\eta_3.$$ Setting
$$1-\dfrac{T_3}{T_2} = 1-\dfrac{T_4}{T_3},$$
we again cancel the 1’s, obtaining
$$-\dfrac{T_3}{T_2} = -\dfrac{T_4}{T_3}.$$
After multiplying by $$-1,$$ we have
$$\dfrac{T_3}{T_2} = \dfrac{T_4}{T_3},$$
which upon cross-multiplication gives
$$T_3^2 = T_2\,T_4.$$
Solving this for $$T_2$$ produces
$$T_2 = \dfrac{T_3^2}{T_4}. \quad -(2)$$
We now substitute the value of $$T_3$$ from equation (1) into equation (2). From (1) we have $$T_3 = \dfrac{T_2^2}{T_1},$$ so its square is
$$T_3^2 = \left(\dfrac{T_2^2}{T_1}\right)^2 = \dfrac{T_2^4}{T_1^2}.$$
Putting this into (2), we get
$$T_2 = \dfrac{\dfrac{T_2^4}{T_1^2}}{T_4}.$$
Simplifying the right-hand side gives
$$T_2 = \dfrac{T_2^4}{T_1^2\,T_4}.$$
To isolate $$T_2,$$ we multiply both sides by $$T_1^2\,T_4:$$
$$(T_1^2\,T_4)\,T_2 = T_2^4.$$
Dividing both sides by $$T_2$$ (which is positive by definition of temperature) yields
$$T_1^2\,T_4 = T_2^3.$$
Taking the cube root of both sides, we arrive at
$$T_2 = (T_1^2\,T_4)^{1/3}.$$
With $$T_2$$ now known, we substitute back into equation (1) to find $$T_3:$$
$$T_3 = \dfrac{T_2^2}{T_1} = \dfrac{\left((T_1^2\,T_4)^{1/3}\right)^2}{T_1}.$$
Because $$\left((T_1^2\,T_4)^{1/3}\right)^2 = (T_1^2\,T_4)^{2/3},$$ we have
$$T_3 = \dfrac{(T_1^2\,T_4)^{2/3}}{T_1}.$$
Splitting the power inside the numerator, $$ (T_1^2\,T_4)^{2/3} = T_1^{4/3}\,T_4^{2/3},$$ so
$$T_3 = \dfrac{T_1^{4/3}\,T_4^{2/3}}{T_1} = T_1^{4/3 - 1}\,T_4^{2/3} = T_1^{1/3}\,T_4^{2/3}.$$
Writing this in compact radical form, we recognize
$$T_3 = (T_1\,T_4^2)^{1/3}.$$
We have therefore obtained
$$T_2 = (T_1^2\,T_4)^{1/3}, \qquad T_3 = (T_1\,T_4^2)^{1/3}.$$
Scanning the given answer choices, we see that these values correspond exactly to Option C.
Hence, the correct answer is Option C.
Create a FREE account and get:
Educational materials for JEE preparation
